QUESTION IMAGE
Question
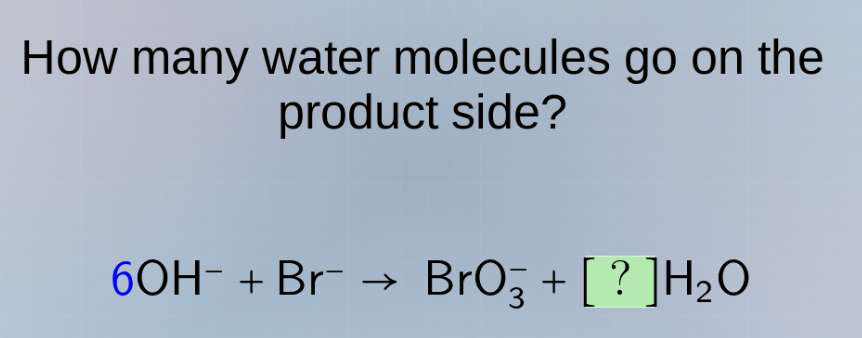
how many water molecules go on the product side? 6oh⁻ + br⁻ → bro₃⁻ + ?h₂o
Step1: Balance oxygen and hydrogen using OH⁻ and H₂O.
We have 6 OH⁻, which provides 6 O and 6 H (from OH⁻: each has 1 O and 1 H).
Step2: Determine H₂O from H and O balance.
The H in 6 OH⁻ is 6, so in H₂O, each H₂O has 2 H. So number of H₂O is $\frac{6}{2}=3$? Wait, no, let's check O. Wait, the product has BrO₃⁻ (3 O) and H₂O. Reactant has 6 OH⁻ (6 O). Let's do balanced redox? Wait, maybe better to balance atoms.
Wait, let's balance the equation properly. Let's consider the reaction: Br⁻ → BrO₃⁻ in basic medium (since OH⁻ is present).
First, balance Br: 1 Br on each side.
Balance O: BrO₃⁻ has 3 O, so add 3 H₂O on left? No, wait, in basic, we use OH⁻ and H₂O. Wait, the given reaction has 6 OH⁻ on left. Let's check H: 6 OH⁻ has 6 H. So on product side, H₂O has H. So number of H₂O: since each H₂O has 2 H, 6 H / 2 H per H₂O = 3? Wait, but let's check O:
Reactant O: 6 (from 6 OH⁻)
Product O: 3 (from BrO₃⁻) + x (from H₂O, x is number of H₂O)
So 6 = 3 + x → x = 3? Wait, no, wait the reaction as given: 6 OH⁻ + Br⁻ → BrO₃⁻ +? H₂O. Wait, maybe I missed electrons. Wait, let's do oxidation state. Br⁻ (oxidation state -1) → BrO₃⁻ (oxidation state +5). So change in oxidation state: +6, so 6 electrons lost.
Now, balance O and H in basic:
Br⁻ → BrO₃⁻
Add 3 H₂O on left to balance O: Br⁻ + 3 H₂O → BrO₃⁻
Add 6 H⁺ on right (but in basic, add 6 OH⁻ to both sides):
Br⁻ + 3 H₂O + 6 OH⁻ → BrO₃⁻ + 6 H⁺ + 6 OH⁻
Simplify: Br⁻ + 3 H₂O + 6 OH⁻ → BrO₃⁻ + 6 H₂O
Then, Br⁻ + 6 OH⁻ → BrO₃⁻ + 3 H₂O + electrons? Wait, no, the electrons: since Br goes from -1 to +5, losing 6 e⁻. So the balanced equation (with electrons) would be Br⁻ + 6 OH⁻ → BrO₃⁻ + 3 H₂O + 6 e⁻. But the given reaction has 6 OH⁻, Br⁻ on left, BrO₃⁻ on right, and H₂O. So from the H balance: 6 OH⁻ has 6 H, so H₂O has 2 H per molecule, so 6 H / 2 = 3 H₂O. And O balance: 6 OH⁻ (6 O) = BrO₃⁻ (3 O) + 3 H₂O (3 O) → 6 O = 3 + 3 = 6. Perfect. So the number of H₂O is 3? Wait, but wait the original equation given is 6 OH⁻ + Br⁻ → BrO₃⁻ +? H₂O. Wait, maybe I made a mistake. Wait, let's count H:
Left side: 6 OH⁻ → 6 H (each OH⁻ has 1 H)
Right side: H₂O has 2 H per molecule. So number of H₂O is 6 H / 2 H per H₂O = 3.
And O: left has 6 (6 OH⁻), right has 3 (BrO₃⁻) + 3 (3 H₂O) = 6. Perfect. So the number of H₂O is 3? Wait, but wait the user's equation: 6 OH⁻ + Br⁻ → BrO₃⁻ +? H₂O. Wait, maybe I missed the other products? Wait, no, maybe the reaction is not balanced for charge. Wait, left charge: 6 OH⁻ (-1 each) + Br⁻ (-1) → total -7. Right charge: BrO₃⁻ (-1) +? H₂O (neutral) → -1. So charge is not balanced. Oh, right! I forgot to balance charge. So Br⁻ → BrO₃⁻: oxidation (losing 6 e⁻). So the charge on left: 6 OH⁻ (-6) + Br⁻ (-1) → -7. On right: BrO₃⁻ (-1) + 3 H₂O (0) + 6 e⁻? Wait, no, the electrons should be on right? Wait, no, in oxidation, electrons are lost, so Br⁻ → BrO₃⁻ + 6 e⁻. Then balance charge with OH⁻. Wait, maybe the correct balanced equation is:
Br⁻ + 6 OH⁻ → BrO₃⁻ + 3 H₂O + 6 e⁻
But the given equation doesn't have electrons, so maybe it's a part of a reaction. But the question is just about the number of H₂O, given 6 OH⁻. So from H balance: 6 OH⁻ has 6 H, so H₂O has 2 H per molecule, so 6/2=3. So the number of H₂O is 3? Wait, but let's check again.
Wait, the equation as written: 6 OH⁻ + Br⁻ → BrO₃⁻ + x H₂O.
H atoms: left: 6 (from 6 OH⁻), right: 2x (from x H₂O). So 6 = 2x → x=3.
O atoms: left: 6 (from 6 OH⁻), right: 3 (from BrO₃⁻) + x (from x H₂O). So 6 = 3 + x → x=3. Perfect. So x=3.
Step3: Confirm the number.
So the number of H₂O molecules is 3. Wait, but wait, maybe I made a mista…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
3