QUESTION IMAGE
Question
- how will you use your periodic table to find the number of neutrons?
- what do an atom, ion and isotope of an element have in common?
- how are atoms, ions, and isotopes different?
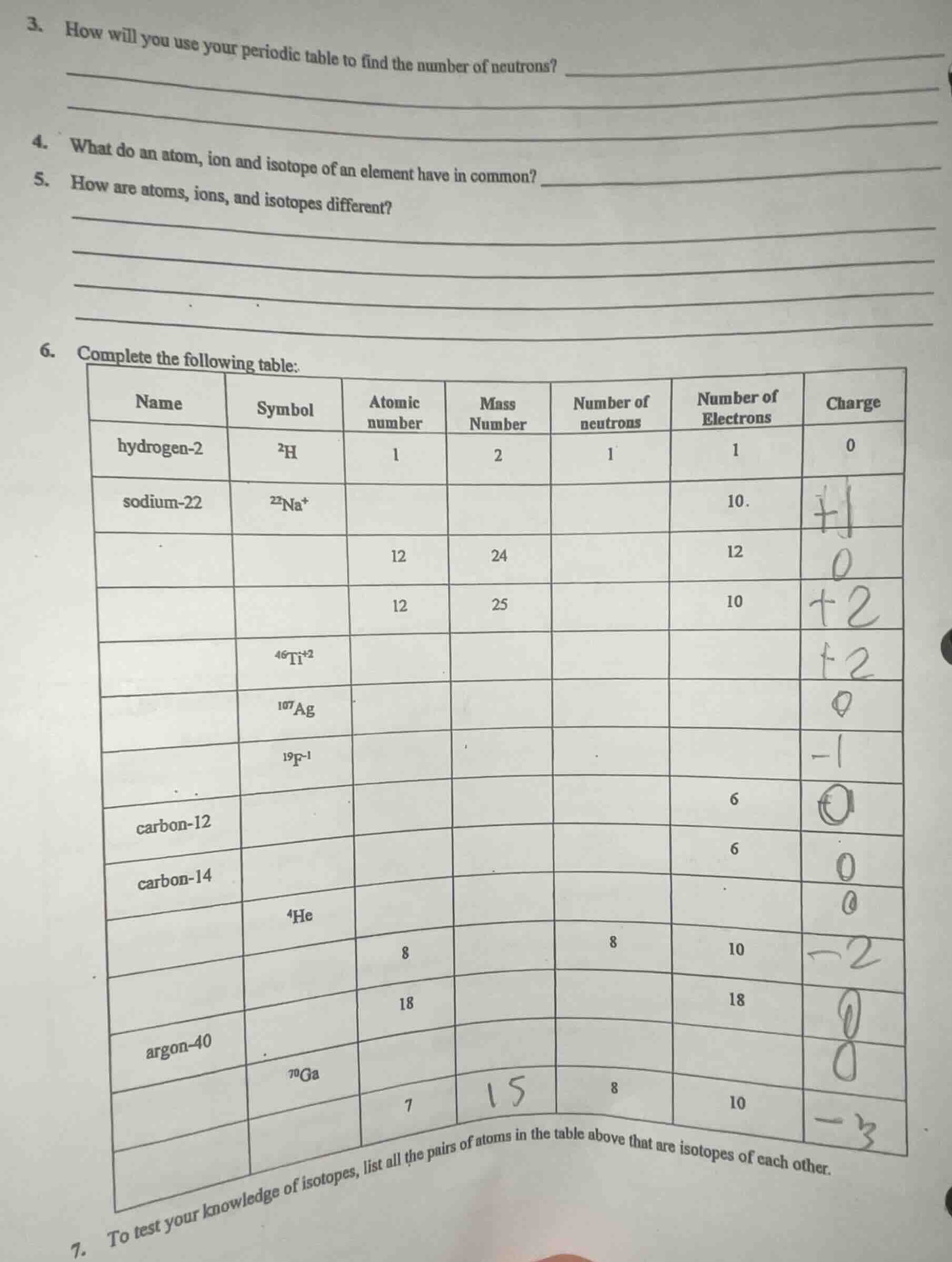
- complete the following table:
| name | symbol | atomic number | mass number | number of neutrons | number of electrons | charge |
|---|---|---|---|---|---|---|
| sodium-22 | $^{22}$na$^+$ | 10. | ||||
| 12 | 24 | 12 | ||||
| 12 | 25 | 10 | ||||
| $^{46}$ti$^{+2}$ | ||||||
| $^{107}$ag | ||||||
| $^{19}$f$^{-1}$ | ||||||
| 6 | ||||||
| carbon-12 | 6 | |||||
| carbon-14 | ||||||
| $^4$he | ||||||
| 8 | 8 | 10 | ||||
| 18 | 18 | |||||
| argon-40 | ||||||
| $^{70}$ga | 8 | |||||
| 7 | 8 | 10 |
- to test your knowledge of isotopes, list all the pairs of atoms in the table above that are isotopes of each other.
Question 3:
To find neutrons, use the periodic table to get the atomic number (protons) and the isotope's mass number. Subtract atomic number from mass number.
Question 4:
All three have the same number of protons, so they belong to the same element (same atomic number).
Question 5:
- Atoms: Neutral, equal protons/electrons.
- Ions: Charged, unequal protons/electrons.
- Isotopes: Same protons, different neutrons/mass numbers.
Question 6:
Use these rules:
- Atomic number = number of protons (from element identity).
- Mass number = protons + neutrons.
- Number of electrons = protons - charge (charge: + for cation, - for anion).
- Charge = protons - electrons.
Question 7:
Isotopes share the same atomic number (same element) but different mass numbers.
---
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
3.
Locate the element's atomic number (protons) on the periodic table, then use the formula:
$\text{Number of neutrons} = \text{Mass number} - \text{Atomic number}$
4.
They all have the same number of protons (same atomic number), meaning they are the same element.
5.
- Atoms are neutral (protons = electrons); ions have a net charge (protons ≠ electrons); isotopes have the same number of protons but different numbers of neutrons (different mass numbers).
6.
| Name | Symbol | Atomic number | Mass Number | Number of neutrons | Number of Electrons | Charge |
|---|---|---|---|---|---|---|
| sodium-22 | $^{22}\text{Na}^+$ | 11 | 22 | 11 | 10 | +1 |
| magnesium-24 | $^{24}\text{Mg}$ | 12 | 24 | 12 | 12 | 0 |
| magnesium-25 cation | $^{25}\text{Mg}^{+2}$ | 12 | 25 | 13 | 10 | +2 |
| titanium-46 cation | $^{46}\text{Ti}^{+2}$ | 22 | 46 | 24 | 20 | +2 |
| silver-107 | $^{107}\text{Ag}$ | 47 | 107 | 60 | 47 | 0 |
| fluorine-19 anion | $^{19}\text{F}^{-1}$ | 9 | 19 | 10 | 10 | -1 |
| carbon-12 | $^{12}\text{C}$ | 6 | 12 | 6 | 6 | 0 |
| carbon-14 | $^{14}\text{C}$ | 6 | 14 | 8 | 6 | 0 |
| helium-4 | $^4\text{He}$ | 2 | 4 | 2 | 2 | 0 |
| oxygen-16 anion | $^{16}\text{O}^{-2}$ | 8 | 16 | 8 | 10 | -2 |
| argon-40 | $^{40}\text{Ar}$ | 18 | 40 | 22 | 18 | 0 |
| gallium-70 | $^{70}\text{Ga}$ | 31 | 70 | 39 | 31 | 0 |
| nitrogen-15 anion | $^{15}\text{N}^{-3}$ | 7 | 15 | 8 | 10 | -3 |
7.
- carbon-12 ($^{12}\text{C}$) and carbon-14 ($^{14}\text{C}$)
- magnesium-24 ($^{24}\text{Mg}$) and magnesium-25 cation ($^{25}\text{Mg}^{+2}$)