QUESTION IMAGE
Question
Question was provided via image upload.
Step1: Recall energy formula
The energy of a photon is $E = hf$, where $h=6.626\times10^{-34}\ \text{J·s}$ (Planck's constant), and $f$ is frequency.
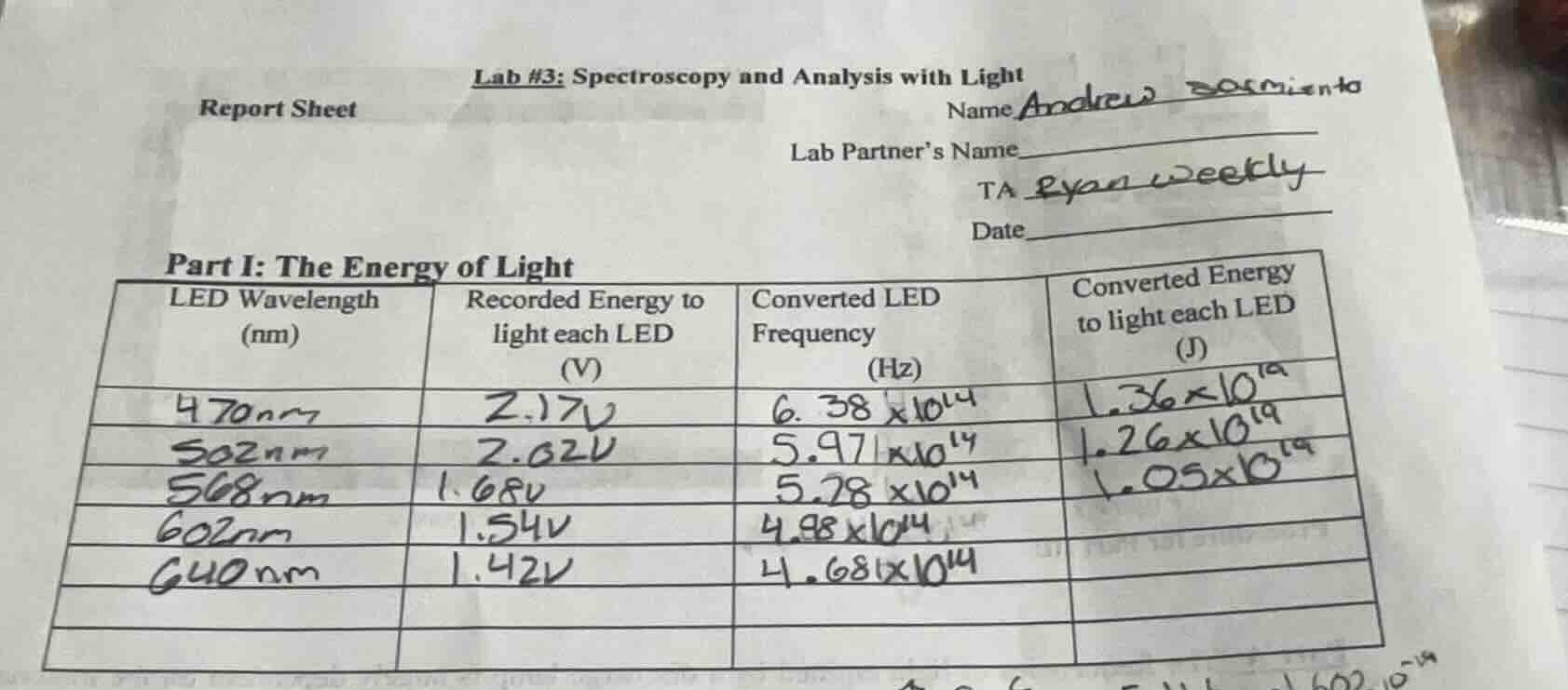
Step2: Calculate for 602 nm LED
Substitute $f=4.98\times10^{14}\ \text{Hz}$
$E = 6.626\times10^{-34} \times 4.98\times10^{14}$
$E \approx 3.30\times10^{-19}\ \text{J}$
Step3: Calculate for 640 nm LED
Substitute $f=4.681\times10^{14}\ \text{Hz}$
$E = 6.626\times10^{-34} \times 4.681\times10^{14}$
$E \approx 3.10\times10^{-19}\ \text{J}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
| LED Wavelength (nm) | Converted Energy to light each LED (J) |
|---|---|
| 640 | $3.10\times10^{-19}$ |