QUESTION IMAGE
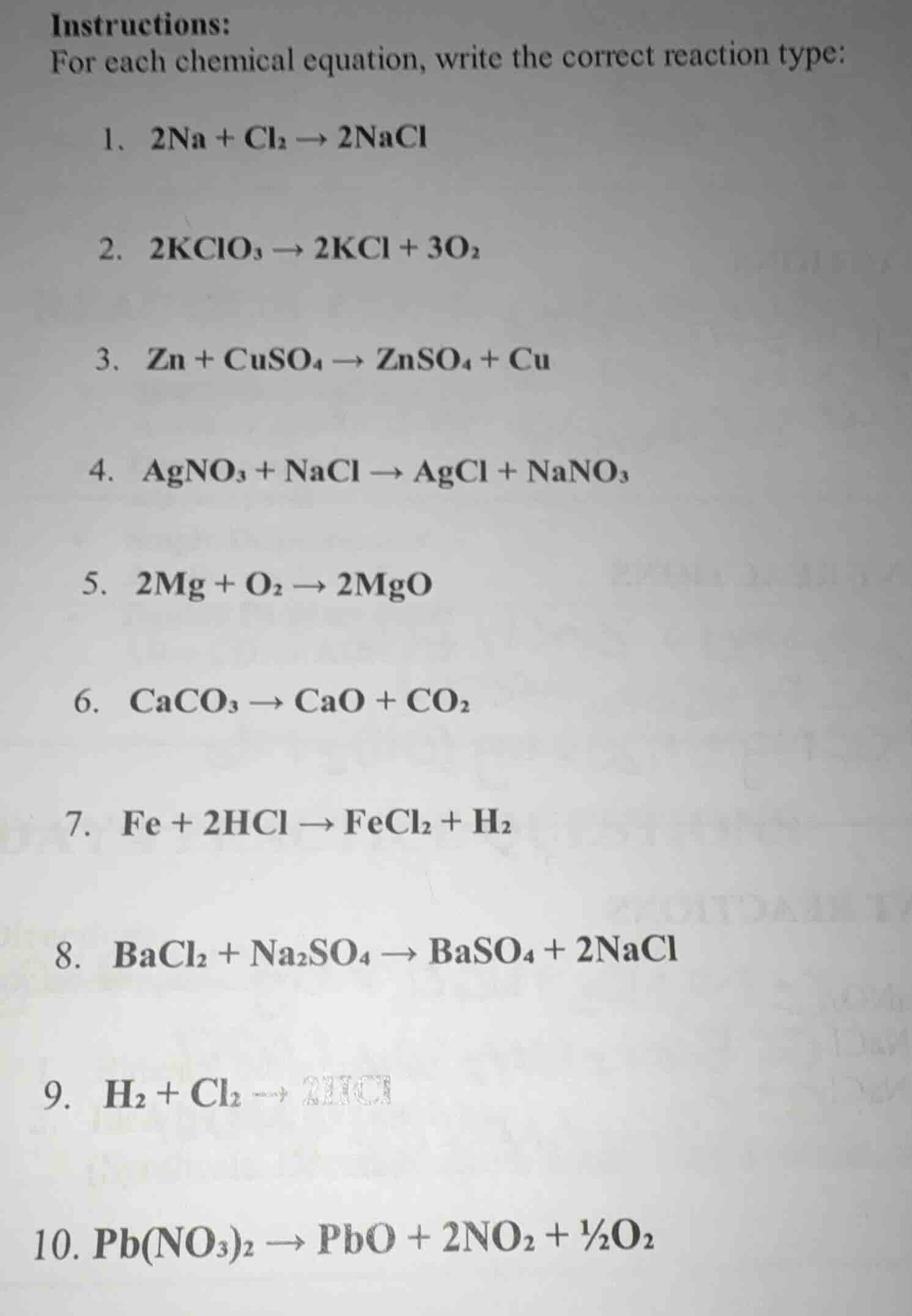
Question
instructions:
for each chemical equation, write the correct reaction type:
- $2\ce{na} + \ce{cl_{2}} \
ightarrow 2\ce{nacl}$
- $2\ce{kclo_{3}} \
ightarrow 2\ce{kcl} + 3\ce{o_{2}}$
- $\ce{zn} + \ce{cuso_{4}} \
ightarrow \ce{znso_{4}} + \ce{cu}$
- $\ce{agno_{3}} + \ce{nacl} \
ightarrow \ce{agcl} + \ce{nano_{3}}$
- $2\ce{mg} + \ce{o_{2}} \
ightarrow 2\ce{mgo}$
- $\ce{caco_{3}} \
ightarrow \ce{cao} + \ce{co_{2}}$
- $\ce{fe} + 2\ce{hcl} \
ightarrow \ce{fecl_{2}} + \ce{h_{2}}$
- $\ce{bacl_{2}} + \ce{na_{2}so_{4}} \
ightarrow \ce{baso_{4}} + 2\ce{nacl}$
- $\ce{h_{2}} + \ce{cl_{2}} \
ightarrow 2\ce{hcl}$
- $\ce{pb(no_{3})_{2}} \
ightarrow \ce{pbo} + 2\ce{no_{2}} + \frac{1}{2}\ce{o_{2}}$
1.
Step1: Identify reaction type
Synthesis (combination) reaction: two or more substances combine to form one product. Here, \(2\text{Na} + \text{Cl}_2
ightarrow 2\text{NaCl}\) has two reactants forming one product.
Step1: Identify reaction type
Decomposition reaction: one compound breaks into two or more products. \(2\text{KClO}_3
ightarrow 2\text{KCl} + 3\text{O}_2\) has one reactant forming two products.
Step1: Identify reaction type
Single - displacement reaction: one element replaces another in a compound. In \(\text{Zn} + \text{CuSO}_4
ightarrow \text{ZnSO}_4 + \text{Cu}\), Zn replaces Cu in \(\text{CuSO}_4\).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Synthesis (Combination) Reaction