QUESTION IMAGE
Question
intermolecular forces
quiz complete
1 2 3 4 5 6 7 8 9 10
time remaining
59:57
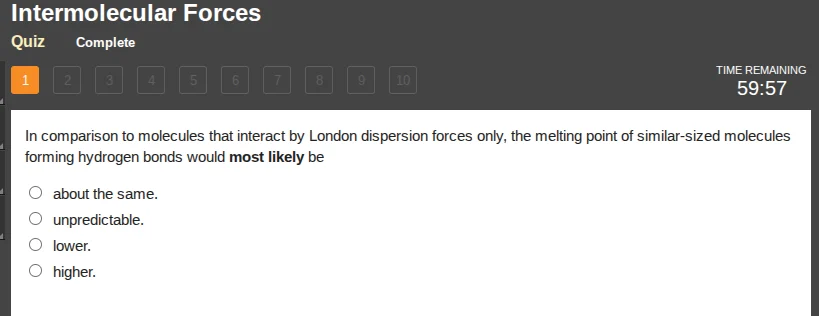
in comparison to molecules that interact by london dispersion forces only, the melting point of similar-sized molecules forming hydrogen bonds would most likely be
about the same.
unpredictable.
lower.
higher.
Hydrogen bonds are stronger intermolecular forces than London dispersion forces. Stronger intermolecular forces require more energy to overcome, so substances with hydrogen bonds have higher melting points than similar-sized substances with only London dispersion forces.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
higher.