QUESTION IMAGE
Question
intermolecular forces
quiz complete
1 2 3 4 5 6 7 8 9 10
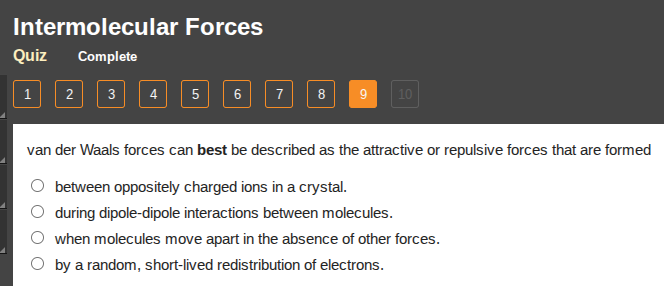
van der waals forces can best be described as the attractive or repulsive forces that are formed
between oppositely charged ions in a crystal.
during dipole-dipole interactions between molecules.
when molecules move apart in the absence of other forces.
by a random, short-lived redistribution of electrons.
Response
Waals component is temporary electron redistribution).
- Option C is incorrect as van der Waals forces include attractive forces that draw molecules together, not just when molecules move apart.
- Option D correctly defines the fundamental origin of London dispersion forces, a key type of van der Waals force, arising from temporary, random electron shifts creating instantaneous dipoles.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Waals component is temporary electron redistribution).
- Option C is incorrect as van der Waals forces include attractive forces that draw molecules together, not just when molecules move apart.
- Option D correctly defines the fundamental origin of London dispersion forces, a key type of van der Waals force, arising from temporary, random electron shifts creating instantaneous dipoles.