QUESTION IMAGE
Question
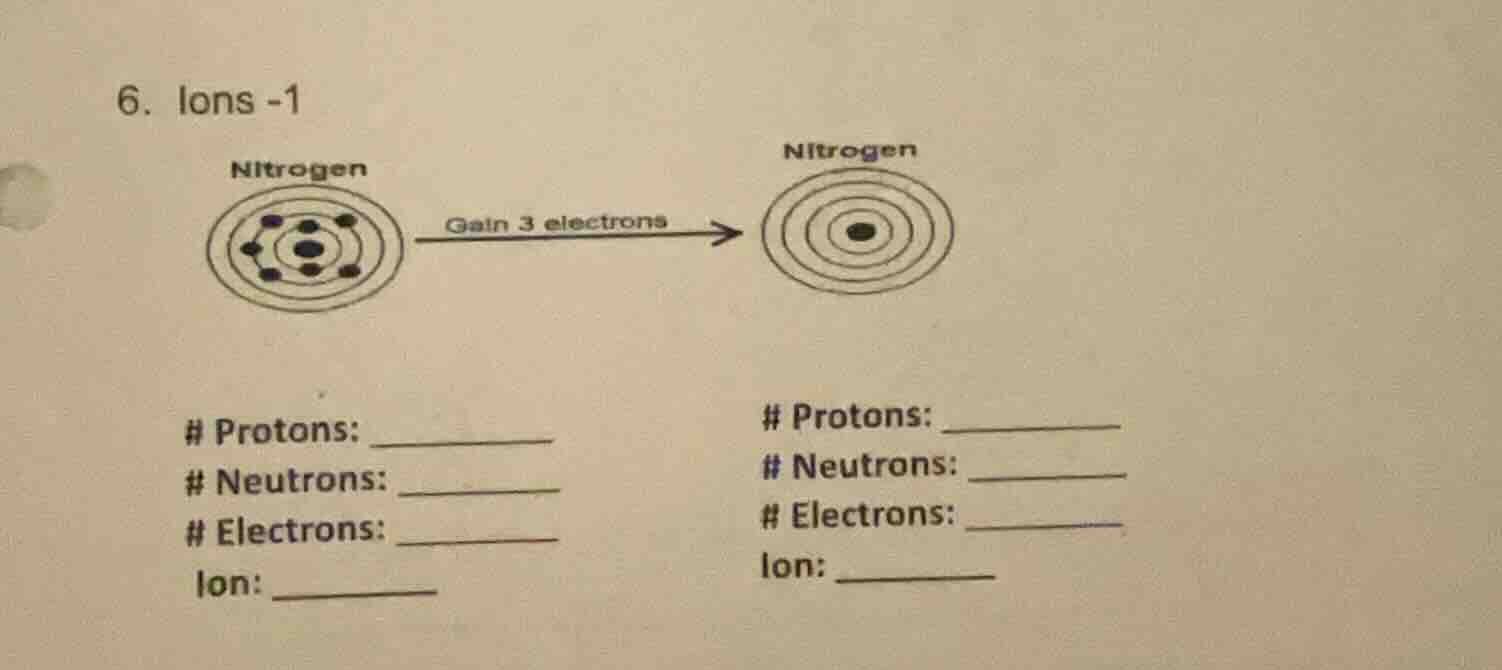
- ions -1
nitrogen
gain 3 electrons
nitrogen
protons: ______
protons: ______
neutrons: ______
neutrons: ______
electrons: ______
electrons: ______
ion: ______
ion: ______
Step1: Find nitrogen's proton count
Nitrogen has an atomic number of 7, so proton count = 7.
Step2: Find neutral nitrogen's electron count
Neutral atoms have equal protons/electrons: 7 electrons.
Step3: Calculate neutron count (common N isotope)
Nitrogen-14 is the most common isotope: neutrons = $14 - 7 = 7$.
Step4: Calculate ion's electron count
Nitrogen gains 3 electrons: $7 + 3 = 10$ electrons.
Step5: Identify ion charge
Gaining 3 electrons gives a -3 charge: $\text{N}^{3-}$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Left (Neutral Nitrogen):
Protons: 7
Neutrons: 7
Electrons: 7
Ion: Neutral (N)
Right (Nitrogen Ion):
Protons: 7
Neutrons: 7
Electrons: 10
Ion: $\text{N}^{3-}$ (Nitride ion)