QUESTION IMAGE
Question
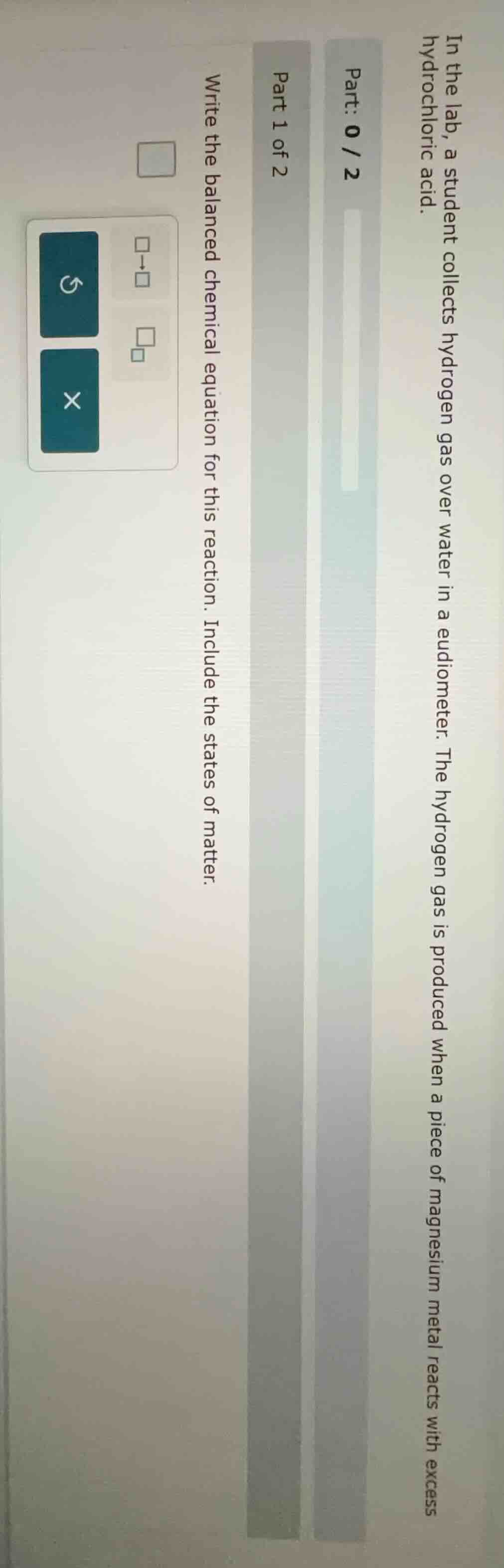
in the lab, a student collects hydrogen gas over water in a eudiometer. the hydrogen gas is produced when a piece of magnesium metal reacts with excess hydrochloric acid. part: 0 / 2 part 1 of 2 write the balanced chemical equation for this reaction. include the states of matter.
Step1: Identify reactants and products
Reactants: Magnesium (Mg, solid) and Hydrochloric acid (HCl, aqueous). Products: Magnesium chloride (MgCl₂, aqueous) and Hydrogen gas (H₂, gas).
Step2: Write unbalanced equation
$ \text{Mg}(s) + \text{HCl}(aq)
ightarrow \text{MgCl}_2(aq) + \text{H}_2(g) $
Step3: Balance Cl and H
There are 2 Cl in MgCl₂, so put 2 in front of HCl. Now H is balanced (2 H in 2 HCl, 2 H in H₂).
$ \text{Mg}(s) + 2\text{HCl}(aq)
ightarrow \text{MgCl}_2(aq) + \text{H}_2(g) $
Step4: Check Mg
Mg is 1 on both sides, so balanced.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$\text{Mg}(s) + 2\text{HCl}(aq)
ightarrow \text{MgCl}_2(aq) + \text{H}_2(g)$