QUESTION IMAGE
Question
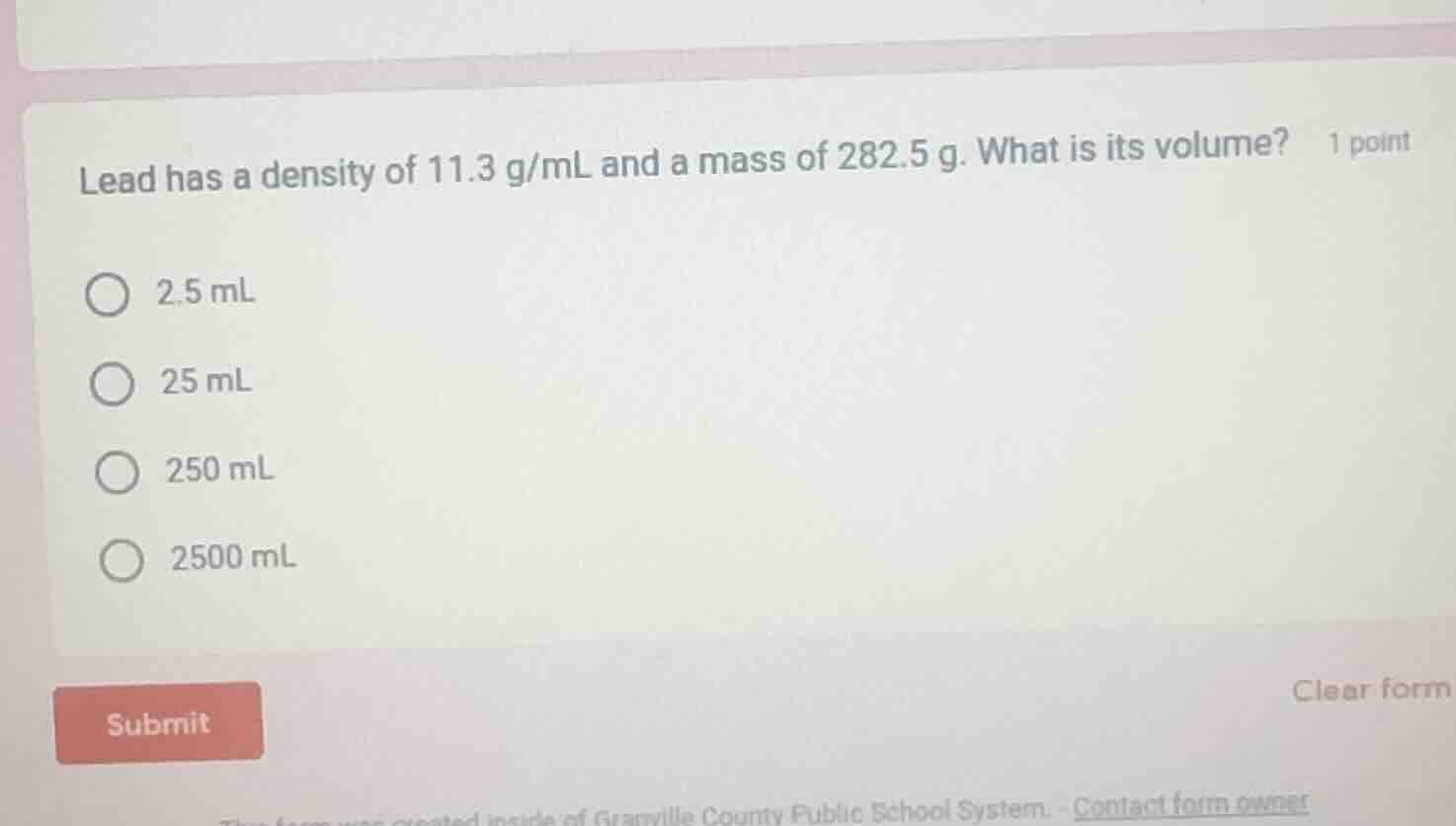
lead has a density of 11.3 g/ml and a mass of 282.5 g. what is its volume? 1 point 2.5 ml 25 ml 250 ml 2500 ml submit clear form
Step1: Recall density formula
The formula for density ($
ho$) is $
ho = \frac{m}{V}$, where $m$ is mass and $V$ is volume. We need to solve for $V$, so rearrange the formula to $V = \frac{m}{
ho}$.
Step2: Substitute values
Given $m = 282.5\ \text{g}$ and $
ho = 11.3\ \text{g/mL}$, substitute into the formula: $V = \frac{282.5}{11.3}$.
Step3: Calculate the division
$\frac{282.5}{11.3} = 25\ \text{mL}$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
25 mL (corresponding to the option "25 mL")