QUESTION IMAGE
Question
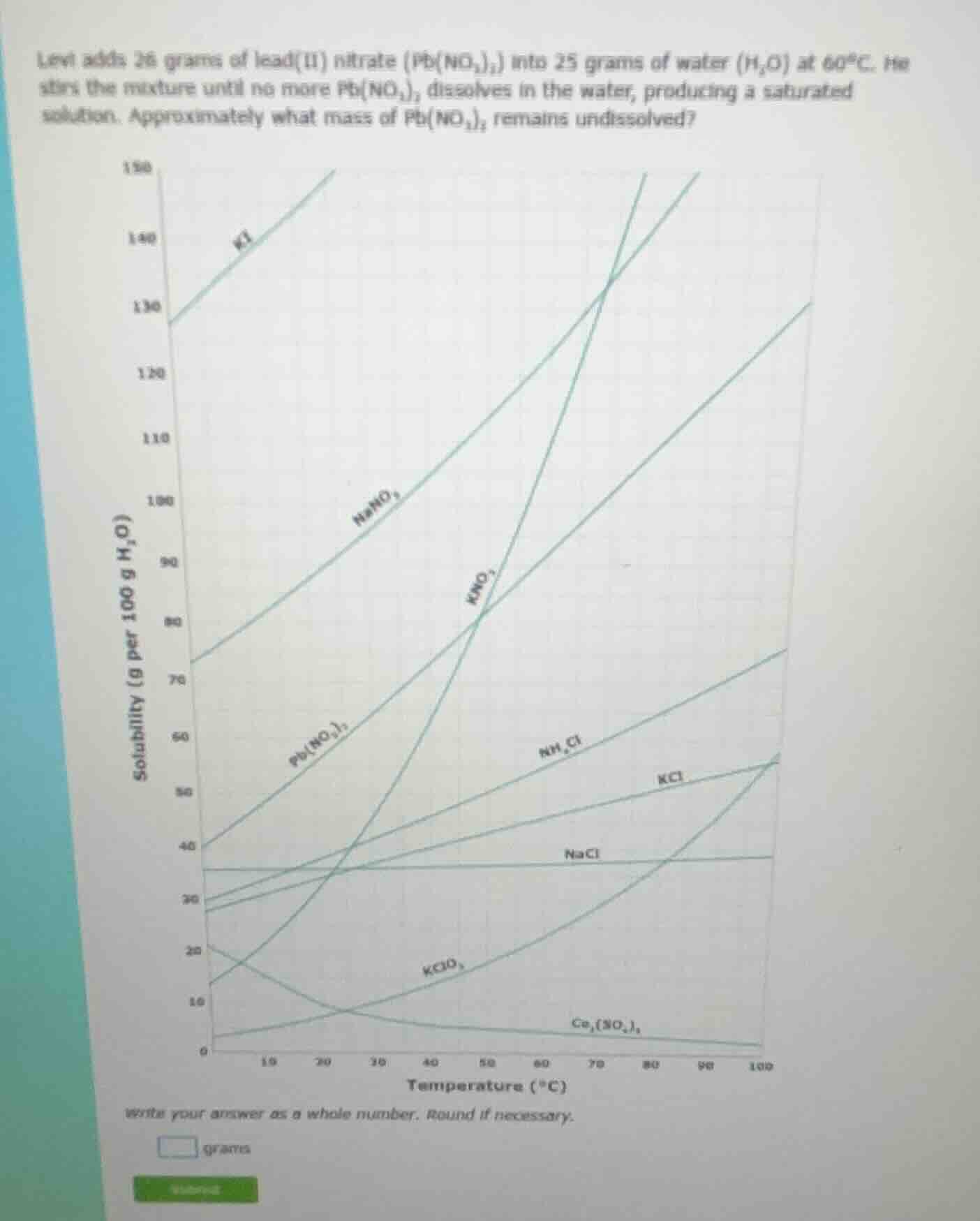
levi adds 26 grams of lead(ii) nitrate (pb(no₃)₂) into 25 grams of water (h₂o) at 60°c. he stirs the mixture until no more pb(no₃)₂ dissolves in the water, producing a saturated solution. approximately what mass of pb(no₃)₂ remains undissolved?
write your answer as a whole number. round if necessary.
______ grams
Step1: Find solubility at 60°C
From the graph, solubility of $\text{Pb(NO}_3\text{)}_2$ at $60^\circ\text{C}$ is $\text{80 g per 100 g } \text{H}_2\text{O}$.
Step2: Calculate dissolved mass in 25g water
Find mass that dissolves in 25g water:
$\text{Dissolved mass} = 80 \times \frac{25}{100} = 20 \text{ g}$
Step3: Calculate undissolved mass
Subtract dissolved mass from added mass:
$\text{Undissolved mass} = 26 - 20 = 6 \text{ g}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
6 grams