QUESTION IMAGE
Question
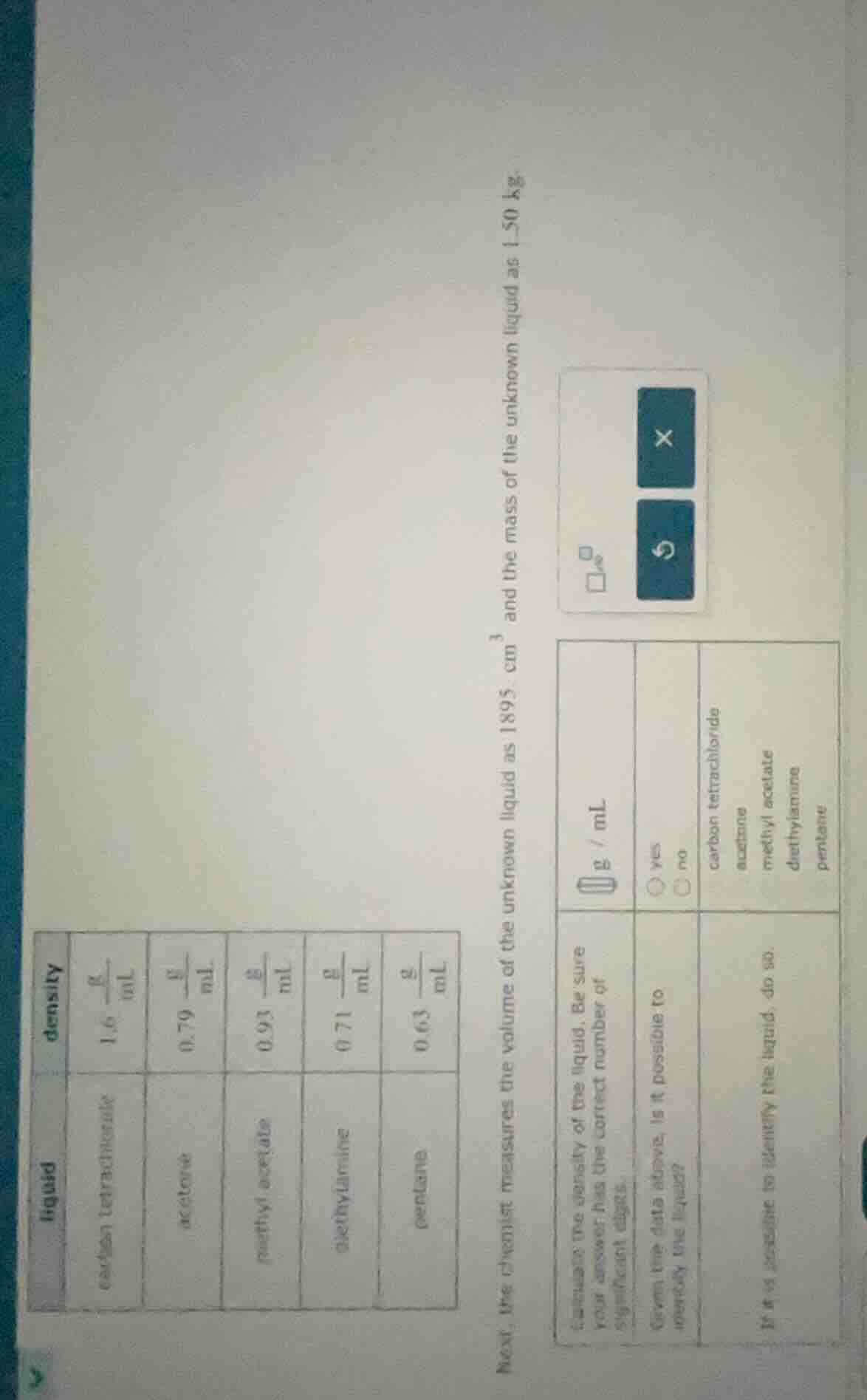
liquid | density
carbon tetrachloride | \\(\frac{1.6\\ \text{g}}{\text{ml}}\\)
acetone | \\(\frac{0.79\\ \text{g}}{\text{ml}}\\)
methyl acetate | \\(\frac{0.93\\ \text{g}}{\text{ml}}\\)
diethylamine | \\(\frac{0.71\\ \text{g}}{\text{ml}}\\)
pentane | \\(\frac{0.63\\ \text{g}}{\text{ml}}\\)
next, the chemist measures the volume of the unknown liquid as 1895 \\(\text{cm}^3\\) and the mass of the unknown liquid as 150 kg.
calculate the density of the liquid. be sure your answer has the correct number of significant digits.
\\(\square\\ \frac{\text{g}}{\text{ml}}\\)
given the data above, is it possible to identify the liquid?
\\(\bigcirc\\) yes \\(\bigcirc\\) no
if it is possible to identify the liquid, do so:
carbon tetrachloride
acetone
methyl acetate
diethylamine
pentane
Step1: Convert mass to grams
$1.50\ \text{kg} = 1.50 \times 1000 = 1500\ \text{g}$
Step2: Convert volume to mL
$1\ \text{cm}^3 = 1\ \text{mL}$, so $1895\ \text{cm}^3 = 1895\ \text{mL}$
Step3: Calculate density
Density formula: $
ho = \frac{m}{V}$
$
ho = \frac{1500\ \text{g}}{1895\ \text{mL}} \approx 0.7915\ \text{g/mL}$
Step4: Round to sig figs
Mass (1.50 kg) has 3 sig figs, volume (1895 cm³) has 4 sig figs. Round to 3 sig figs: $0.792\ \text{g/mL} \approx 0.79\ \text{g/mL}$
Step5: Match density to liquid
$0.79\ \text{g/mL}$ matches acetone's density.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Calculated density: $\boldsymbol{0.79\ \text{g/mL}}$
Is it possible to identify the liquid? $\boldsymbol{\text{yes}}$
Identified liquid: $\boldsymbol{\text{acetone}}$