QUESTION IMAGE
Question
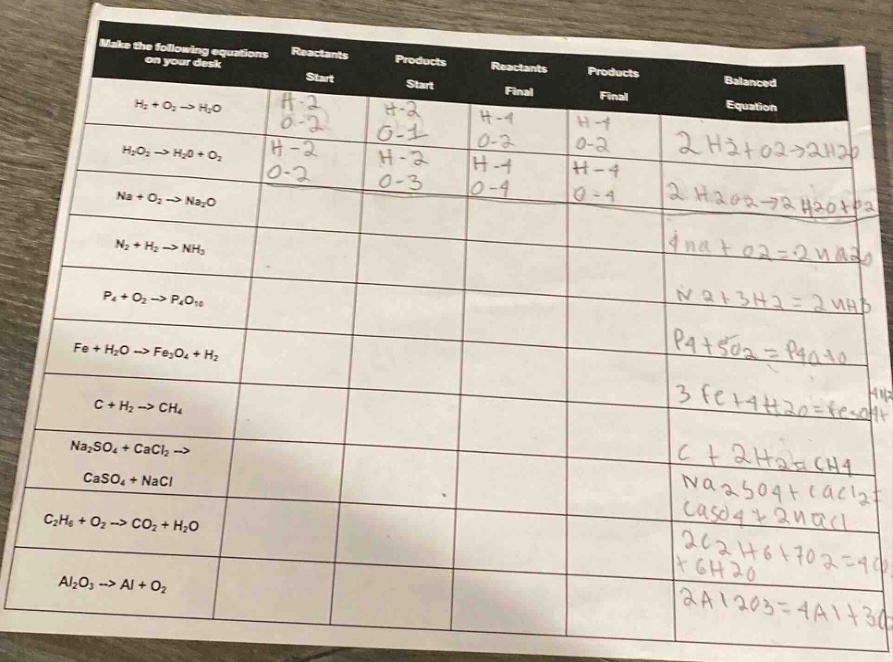
make the following equations on your desk reactants start products start reactants final products final balanced equation h₂ + o₂ → h₂o h-2 o-2 h-2 o-1 h-4 o-2 h-4 o-2 2 h₂ + o₂ → 2 h₂o h₂o₂ → h₂o + o₂ h-2 o-2 h-2 o-3 h-4 o-4 h-4 o-4 2 h₂o₂ → 2 h₂o + o₂ na + o₂ → na₂o 4 na + o₂ → 2 na₂o n₂ + h₂ → nh₃ n₂ + 3 h₂ → 2 nh₃ p₄ + o₂ → p₄o₁₀ p₄ + 5 o₂ → p₄o₁₀ fe + h₂o → fe₃o₄ + h₂ 3 fe + 4 h₂o → fe₃o₄ + 4 h₂ c + h₂ → ch₄ c + 2 h₂ → ch₄ na₂so₄ + cacl₂ → caso₄ + nacl na₂so₄ + cacl₂ → caso₄ + 2 nacl c₂h₆ + o₂ → co₂ + h₂o 2 c₂h₆ + 7 o₂ → 4 co₂ + 6 h₂o al₂o₃ → al + o₂ 2 al₂o₃ → 4 al + 3 o₂
To balance the chemical equations, we follow these steps for each:
1. $\boldsymbol{H_2 + O_2
ightarrow H_2O}$
- Step 1: Count atoms on each side.
Reactants: $H = 2$, $O = 2$; Products: $H = 2$, $O = 1$.
- Step 2: Balance O first.
Multiply $H_2O$ by 2: $H_2 + O_2
ightarrow 2H_2O$. Now $O = 2$ (balanced), but $H = 4$ (unbalanced).
- Step 3: Balance H.
Multiply $H_2$ by 2: $2H_2 + O_2
ightarrow 2H_2O$.
Final check: Reactants ($H=4$, $O=2$); Products ($H=4$, $O=2$). Balanced.
2. $\boldsymbol{H_2O_2
ightarrow H_2O + O_2}$
- Step 1: Count atoms.
Reactants: $H = 2$, $O = 2$; Products: $H = 2$, $O = 3$ (unbalanced).
- Step 2: Balance O.
Multiply $H_2O_2$ by 2 and $H_2O$ by 2: $2H_2O_2
ightarrow 2H_2O + O_2$.
Check: Reactants ($H=4$, $O=4$); Products ($H=4$, $O=4$). Balanced.
3. $\boldsymbol{Na + O_2
ightarrow Na_2O}$
- Step 1: Count atoms.
Reactants: $Na = 1$, $O = 2$; Products: $Na = 2$, $O = 1$ (unbalanced).
- Step 2: Balance Na first.
Multiply $Na$ by 2: $2Na + O_2
ightarrow Na_2O$. Now $Na = 2$ (balanced), but $O = 2$ (unbalanced, product has $O=1$).
- Step 3: Balance O.
Multiply $Na_2O$ by 2: $2Na + O_2
ightarrow 2Na_2O$. Now $Na = 4$ (unbalanced).
- Step 4: Adjust Na.
Multiply $Na$ by 4: $4Na + O_2
ightarrow 2Na_2O$.
Check: Reactants ($Na=4$, $O=2$); Products ($Na=4$, $O=2$). Balanced.
4. $\boldsymbol{N_2 + H_2
ightarrow NH_3}$
- Step 1: Count atoms.
Reactants: $N = 2$, $H = 2$; Products: $N = 1$, $H = 3$ (unbalanced).
- Step 2: Balance N first.
Multiply $NH_3$ by 2: $N_2 + H_2
ightarrow 2NH_3$. Now $N = 2$ (balanced), but $H = 6$ (unbalanced).
- Step 3: Balance H.
Multiply $H_2$ by 3: $N_2 + 3H_2
ightarrow 2NH_3$.
Check: Reactants ($N=2$, $H=6$); Products ($N=2$, $H=6$). Balanced.
5. $\boldsymbol{P_4 + O_2
ightarrow P_4O_{10}}$
- Step 1: Count atoms.
Reactants: $P = 4$, $O = 2$; Products: $P = 4$, $O = 10$ (unbalanced).
- Step 2: Balance O.
Multiply $O_2$ by 5: $P_4 + 5O_2
ightarrow P_4O_{10}$.
Check: Reactants ($P=4$, $O=10$); Products ($P=4$, $O=10$). Balanced.
6. $\boldsymbol{Fe + H_2O
ightarrow Fe_3O_4 + H_2}$
- Step 1: Count atoms.
Reactants: $Fe = 1$, $H = 2$, $O = 1$; Products: $Fe = 3$, $H = 2$, $O = 4$ (unbalanced).
- Step 2: Balance Fe first.
Multiply $Fe$ by 3: $3Fe + H_2O
ightarrow Fe_3O_4 + H_2$. Now $Fe = 3$ (balanced), but $O = 4$ (unbalanced, reactant has $O=1$).
- Step 3: Balance O.
Multiply $H_2O$ by 4: $3Fe + 4H_2O
ightarrow Fe_3O_4 + H_2$. Now $O = 4$ (balanced), but $H = 8$ (unbalanced, product has $H=2$).
- Step 4: Balance H.
Multiply $H_2$ by 4: $3Fe + 4H_2O
ightarrow Fe_3O_4 + 4H_2$.
Check: Reactants ($Fe=3$, $H=8$, $O=4$); Products ($Fe=3$, $H=8$, $O=4$). Balanced.
7. $\boldsymbol{C + H_2
ightarrow CH_4}$
- Step 1: Count atoms.
Reactants: $C = 1$, $H = 2$; Products: $C = 1$, $H = 4$ (unbalanced).
- Step 2: Balance H.
Multiply $H_2$ by 2: $C + 2H_2
ightarrow CH_4$.
Check: Reactants ($C=1$, $H=4$); Products ($C=1$, $H=4$). Balanced.
8. $\boldsymbol{Na_2SO_4 + CaCl_2
ightarrow CaSO_4 + NaCl}$
- Step 1: Count atoms.
Reactants: $Na = 2$, $S = 1$, $O = 4$, $Ca = 1$, $Cl = 2$; Products: $Na = 1$, $S = 1$, $O = 4$, $Ca = 1$, $Cl = 1$ (unbalanced).
- Step 2: Balance Na and Cl.
Multiply $NaCl$ by 2: $Na_2SO_4 + CaCl_2
ightarrow CaSO_4 + 2NaCl$.
Check: Reactants ($Na=2$, $S=1$, $O=4$, $Ca=1$, $Cl=2$); Products ($Na=2$, $S=1$, $O=4$, $Ca=1$, $Cl=2$). Balanced.
9. $\boldsymbol{C_2H_6 + O_2
ightarrow CO_2 + H_2O}$
- **Step 1: Count at…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
To balance the chemical equations, we follow these steps for each:
1. $\boldsymbol{H_2 + O_2
ightarrow H_2O}$
- Step 1: Count atoms on each side.
Reactants: $H = 2$, $O = 2$; Products: $H = 2$, $O = 1$.
- Step 2: Balance O first.
Multiply $H_2O$ by 2: $H_2 + O_2
ightarrow 2H_2O$. Now $O = 2$ (balanced), but $H = 4$ (unbalanced).
- Step 3: Balance H.
Multiply $H_2$ by 2: $2H_2 + O_2
ightarrow 2H_2O$.
Final check: Reactants ($H=4$, $O=2$); Products ($H=4$, $O=2$). Balanced.
2. $\boldsymbol{H_2O_2
ightarrow H_2O + O_2}$
- Step 1: Count atoms.
Reactants: $H = 2$, $O = 2$; Products: $H = 2$, $O = 3$ (unbalanced).
- Step 2: Balance O.
Multiply $H_2O_2$ by 2 and $H_2O$ by 2: $2H_2O_2
ightarrow 2H_2O + O_2$.
Check: Reactants ($H=4$, $O=4$); Products ($H=4$, $O=4$). Balanced.
3. $\boldsymbol{Na + O_2
ightarrow Na_2O}$
- Step 1: Count atoms.
Reactants: $Na = 1$, $O = 2$; Products: $Na = 2$, $O = 1$ (unbalanced).
- Step 2: Balance Na first.
Multiply $Na$ by 2: $2Na + O_2
ightarrow Na_2O$. Now $Na = 2$ (balanced), but $O = 2$ (unbalanced, product has $O=1$).
- Step 3: Balance O.
Multiply $Na_2O$ by 2: $2Na + O_2
ightarrow 2Na_2O$. Now $Na = 4$ (unbalanced).
- Step 4: Adjust Na.
Multiply $Na$ by 4: $4Na + O_2
ightarrow 2Na_2O$.
Check: Reactants ($Na=4$, $O=2$); Products ($Na=4$, $O=2$). Balanced.
4. $\boldsymbol{N_2 + H_2
ightarrow NH_3}$
- Step 1: Count atoms.
Reactants: $N = 2$, $H = 2$; Products: $N = 1$, $H = 3$ (unbalanced).
- Step 2: Balance N first.
Multiply $NH_3$ by 2: $N_2 + H_2
ightarrow 2NH_3$. Now $N = 2$ (balanced), but $H = 6$ (unbalanced).
- Step 3: Balance H.
Multiply $H_2$ by 3: $N_2 + 3H_2
ightarrow 2NH_3$.
Check: Reactants ($N=2$, $H=6$); Products ($N=2$, $H=6$). Balanced.
5. $\boldsymbol{P_4 + O_2
ightarrow P_4O_{10}}$
- Step 1: Count atoms.
Reactants: $P = 4$, $O = 2$; Products: $P = 4$, $O = 10$ (unbalanced).
- Step 2: Balance O.
Multiply $O_2$ by 5: $P_4 + 5O_2
ightarrow P_4O_{10}$.
Check: Reactants ($P=4$, $O=10$); Products ($P=4$, $O=10$). Balanced.
6. $\boldsymbol{Fe + H_2O
ightarrow Fe_3O_4 + H_2}$
- Step 1: Count atoms.
Reactants: $Fe = 1$, $H = 2$, $O = 1$; Products: $Fe = 3$, $H = 2$, $O = 4$ (unbalanced).
- Step 2: Balance Fe first.
Multiply $Fe$ by 3: $3Fe + H_2O
ightarrow Fe_3O_4 + H_2$. Now $Fe = 3$ (balanced), but $O = 4$ (unbalanced, reactant has $O=1$).
- Step 3: Balance O.
Multiply $H_2O$ by 4: $3Fe + 4H_2O
ightarrow Fe_3O_4 + H_2$. Now $O = 4$ (balanced), but $H = 8$ (unbalanced, product has $H=2$).
- Step 4: Balance H.
Multiply $H_2$ by 4: $3Fe + 4H_2O
ightarrow Fe_3O_4 + 4H_2$.
Check: Reactants ($Fe=3$, $H=8$, $O=4$); Products ($Fe=3$, $H=8$, $O=4$). Balanced.
7. $\boldsymbol{C + H_2
ightarrow CH_4}$
- Step 1: Count atoms.
Reactants: $C = 1$, $H = 2$; Products: $C = 1$, $H = 4$ (unbalanced).
- Step 2: Balance H.
Multiply $H_2$ by 2: $C + 2H_2
ightarrow CH_4$.
Check: Reactants ($C=1$, $H=4$); Products ($C=1$, $H=4$). Balanced.
8. $\boldsymbol{Na_2SO_4 + CaCl_2
ightarrow CaSO_4 + NaCl}$
- Step 1: Count atoms.
Reactants: $Na = 2$, $S = 1$, $O = 4$, $Ca = 1$, $Cl = 2$; Products: $Na = 1$, $S = 1$, $O = 4$, $Ca = 1$, $Cl = 1$ (unbalanced).
- Step 2: Balance Na and Cl.
Multiply $NaCl$ by 2: $Na_2SO_4 + CaCl_2
ightarrow CaSO_4 + 2NaCl$.
Check: Reactants ($Na=2$, $S=1$, $O=4$, $Ca=1$, $Cl=2$); Products ($Na=2$, $S=1$, $O=4$, $Ca=1$, $Cl=2$). Balanced.
9. $\boldsymbol{C_2H_6 + O_2
ightarrow CO_2 + H_2O}$
- Step 1: Count atoms.
Reactants: $C = 2$, $H = 6$, $O = 2$; Products: $C = 1$, $H = 2$, $O = 3$ (unbalanced).
- Step 2: Balance C first.
Multiply $CO_2$ by 2: $C_2H_6 + O_2
ightarrow 2CO_2 + H_2O$. Now $C = 2$ (balanced), but $H = 6$ (unbalanced, product has $H=2$).
- Step 3: Balance H.
Multiply $H_2O$ by 3: $C_2H_6 + O_2
ightarrow 2CO_2 + 3H_2O$. Now $H = 6$ (balanced), but $O = 7$ (unbalanced, reactant has $O=2$).
- Step 4: Balance O.
Multiply $O_2$ by $\frac{7}{2}$ (or use whole numbers by multiplying all by 2):
$2C_2H_6 + 7O_2
ightarrow 4CO_2 + 6H_2O$.
Check: Reactants ($C=4$, $H=12$, $O=14$); Products ($C=4$, $H=12$, $O=14$). Balanced.
10. $\boldsymbol{Al_2O_3
ightarrow Al + O_2}$
- Step 1: Count atoms.
Reactants: $Al = 2$, $O = 3$; Products: $Al = 1$, $O = 2$ (unbalanced).
- Step 2: Balance Al first.
Multiply $Al$ by 2: $Al_2O_3
ightarrow 2Al + O_2$. Now $Al = 2$ (balanced), but $O = 3$ (unbalanced, product has $O=2$).
- Step 3: Balance O (use least common multiple of 3 and 2, which is 6).
Multiply $Al_2O_3$ by 2 and $O_2$ by 3: $2Al_2O_3
ightarrow 4Al + 3O_2$.
Check: Reactants ($Al=4$, $O=6$); Products ($Al=4$, $O=6$). Balanced.
Final Balanced Equations:
- $2H_2 + O_2
ightarrow 2H_2O$
- $2H_2O_2
ightarrow 2H_2O + O_2$
- $4Na + O_2
ightarrow 2Na_2O$
- $N_2 + 3H_2
ightarrow 2NH_3$
- $P_4 + 5O_2
ightarrow P_4O_{10}$
- $3Fe + 4H_2O
ightarrow Fe_3O_4 + 4H_2$
- $C + 2H_2
ightarrow CH_4$
- $Na_2SO_4 + CaCl_2
ightarrow CaSO_4 + 2NaCl$
- $2C_2H_6 + 7O_2
ightarrow 4CO_2 + 6H_2O$
- $2Al_2O_3
ightarrow 4Al + 3O_2$