QUESTION IMAGE
Question
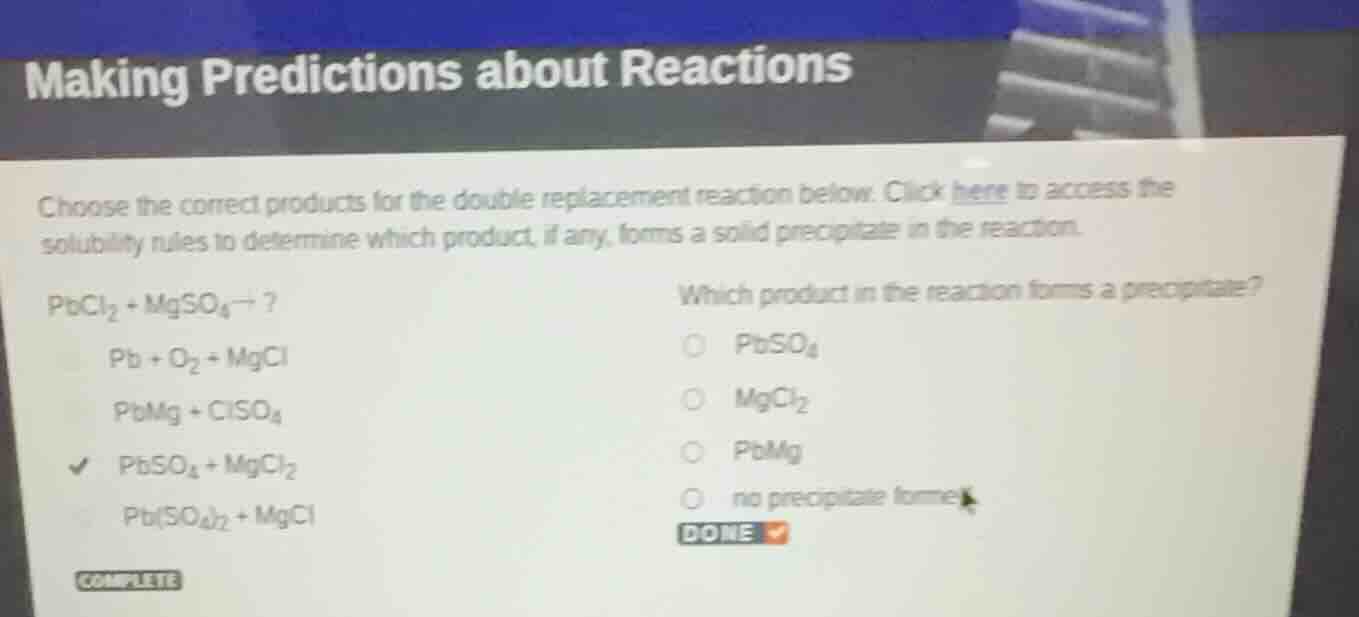
making predictions about reactions
choose the correct products for the double replacement reaction below. click here to access the solubility rules to determine which product, if any, forms a solid precipitate in the reaction.
$\text{pbcl}_2 + \text{mgso}_4
ightarrow ?$
$\text{pb} + \text{o}_2 + \text{mgcl}$
$\text{pbmg} + \text{clso}_4$
$\text{pbso}_4 + \text{mgcl}_2$
$\text{pb(so}_4\text{)}_2 + \text{mgcl}$
which product in the reaction forms a precipitate?
$\text{pbso}_4$
$\text{mgcl}_2$
$\text{pbmg}$
no precipitate forms
done
complete
- For double replacement reactions, cations and anions swap partners. For $\text{PbCl}_2 + \text{MgSO}_4$, lead(II) pairs with sulfate, and magnesium pairs with chloride, forming $\text{PbSO}_4 + \text{MgCl}_2$.
- Using solubility rules: Most sulfate salts are soluble, but $\text{PbSO}_4$ is an insoluble exception. $\text{MgCl}_2$ is a soluble chloride salt. Thus, $\text{PbSO}_4$ forms a precipitate.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Correct reaction products: $\text{PbSO}_4 + \text{MgCl}_2$
Precipitate-forming product: $\text{PbSO}_4$