QUESTION IMAGE
Question
mass number equals
the number of
________ + ________
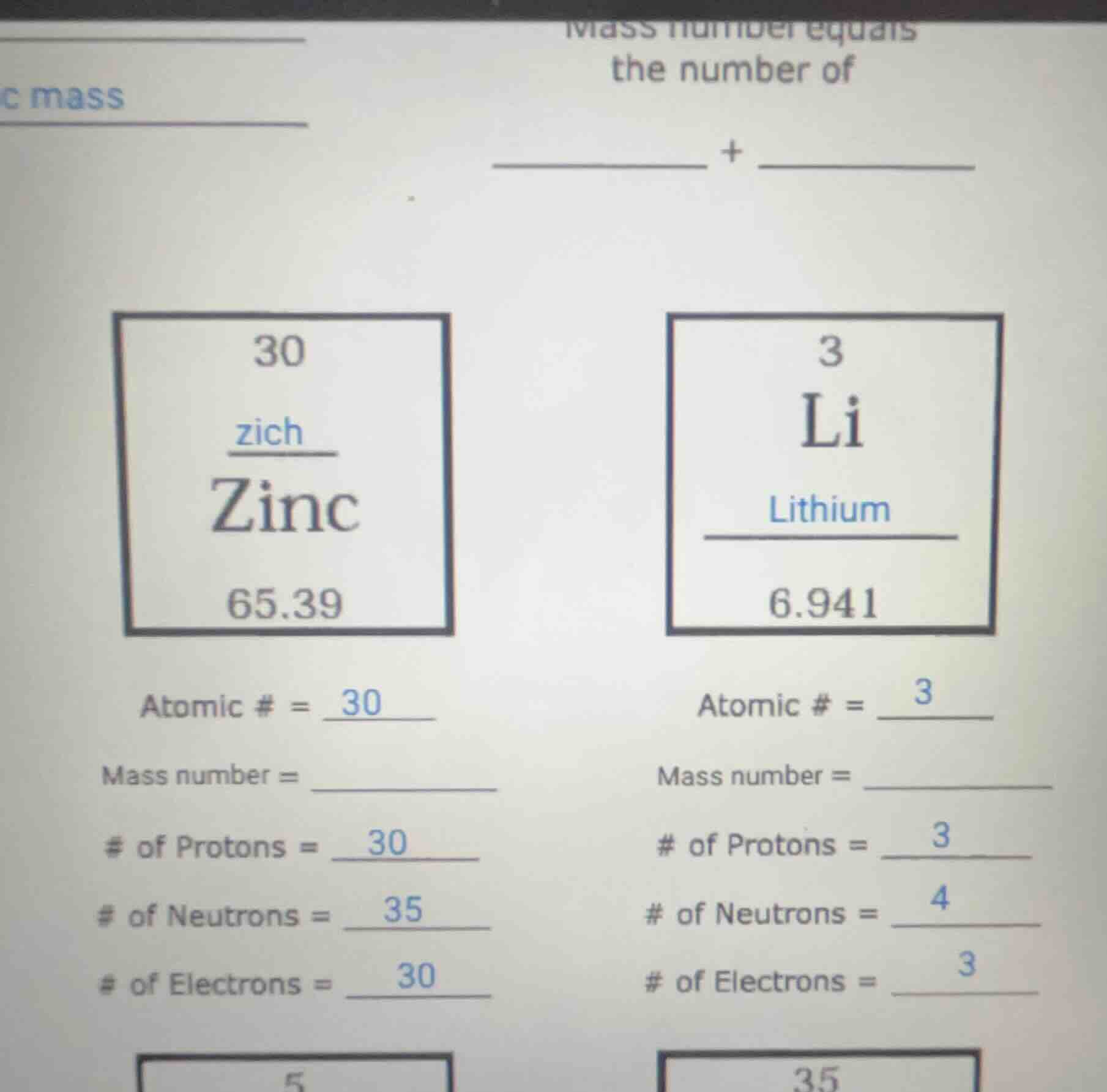
30
zich
zinc
65.39
atomic # = 30
mass number = __________
of protons = 30
of neutrons = 35
of electrons = 30
3
li
lithium
6.941
atomic # = 3
mass number = __________
of protons = 3
of neutrons = 4
of electrons = 3
Step1: Recall mass number formula
The mass number of an atom is defined as the sum of the number of protons and neutrons in its nucleus:
$$\text{Mass number} = \text{Number of Protons} + \text{Number of Neutrons}$$
Step2: Calculate Zinc's mass number
For Zinc, number of protons = 30, number of neutrons = 35. Substitute values:
$$\text{Mass number (Zinc)} = 30 + 35 = 65$$
Step3: Calculate Lithium's mass number
For Lithium, number of protons = 3, number of neutrons = 4. Substitute values:
$$\text{Mass number (Lithium)} = 3 + 4 = 7$$
Step4: Fill top formula blanks
The mass number equals protons plus neutrons.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Top blanks (left to right): Protons, Neutrons
Zinc: Mass number = 65
Lithium: Mass number = 7