QUESTION IMAGE
Question
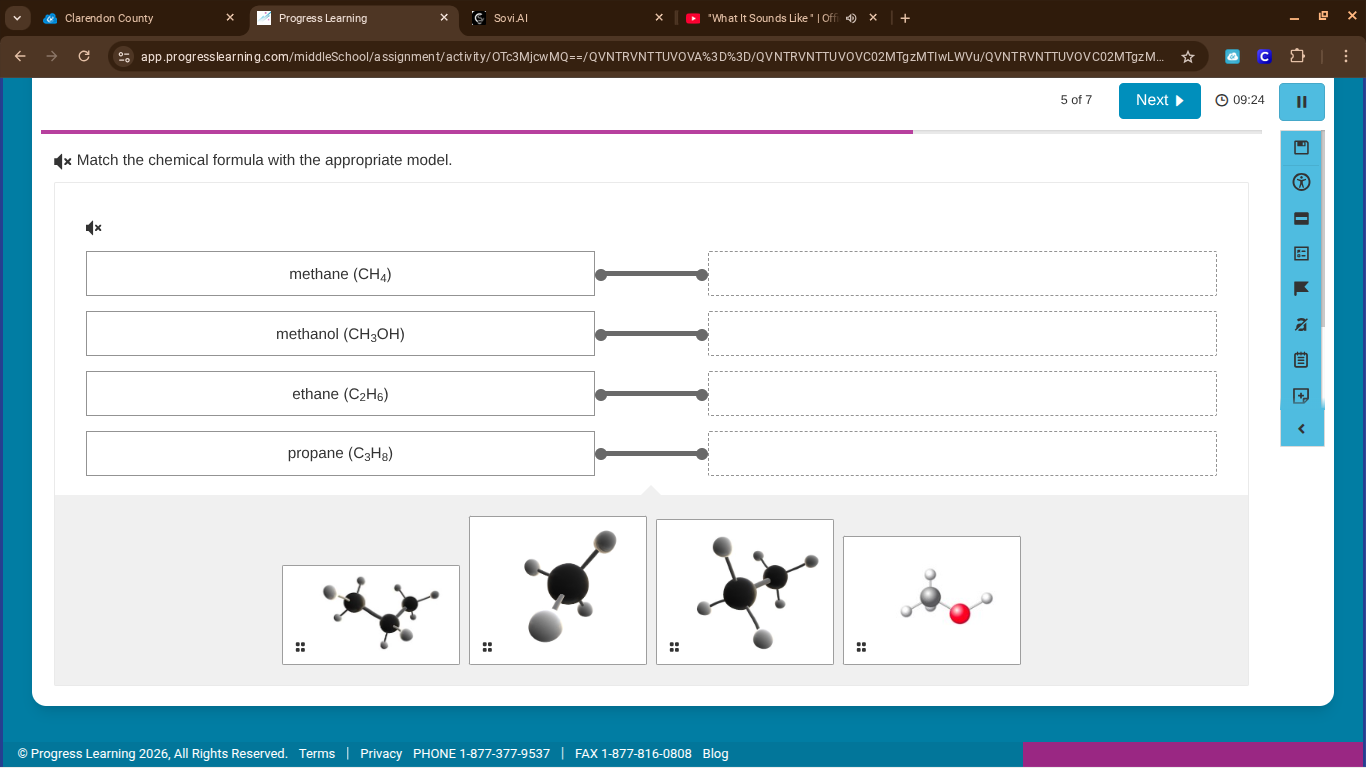
match the chemical formula with the appropriate model.
methane (ch₄)
methanol (ch₃oh)
ethane (c₂h₆)
propane (c₃h₈)
To solve this problem, we analyze the molecular structure of each compound:
Methane ($\boldsymbol{CH_4}$)
Methane has 1 carbon atom bonded to 4 hydrogen atoms. The second model (black sphere with 4 smaller spheres) matches this structure (1 central C, 4 H).
Methanol ($\boldsymbol{CH_3OH}$)
Methanol has 1 carbon, 3 hydrogens, 1 oxygen, and 1 hydrogen bonded to oxygen. The fourth model (gray sphere with a red sphere and two small spheres) matches (C - $CH_3$, O - $OH$).
Ethane ($\boldsymbol{C_2H_6}$)
Ethane has 2 carbon atoms bonded to each other, with 3 hydrogens per carbon. The third model (two black spheres, each with hydrogens) matches (2 C atoms, 6 H atoms).
Propane ($\boldsymbol{C_3H_8}$)
Propane has 3 carbon atoms in a chain, with hydrogens attached. The first model (three black spheres in a chain, with hydrogens) matches (3 C atoms, 8 H atoms).
Final Matches:
- Methane ($CH_4$) → Second model
- Methanol ($CH_3OH$) → Fourth model
- Ethane ($C_2H_6$) → Third model
- Propane ($C_3H_8$) → First model
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
To solve this problem, we analyze the molecular structure of each compound:
Methane ($\boldsymbol{CH_4}$)
Methane has 1 carbon atom bonded to 4 hydrogen atoms. The second model (black sphere with 4 smaller spheres) matches this structure (1 central C, 4 H).
Methanol ($\boldsymbol{CH_3OH}$)
Methanol has 1 carbon, 3 hydrogens, 1 oxygen, and 1 hydrogen bonded to oxygen. The fourth model (gray sphere with a red sphere and two small spheres) matches (C - $CH_3$, O - $OH$).
Ethane ($\boldsymbol{C_2H_6}$)
Ethane has 2 carbon atoms bonded to each other, with 3 hydrogens per carbon. The third model (two black spheres, each with hydrogens) matches (2 C atoms, 6 H atoms).
Propane ($\boldsymbol{C_3H_8}$)
Propane has 3 carbon atoms in a chain, with hydrogens attached. The first model (three black spheres in a chain, with hydrogens) matches (3 C atoms, 8 H atoms).
Final Matches:
- Methane ($CH_4$) → Second model
- Methanol ($CH_3OH$) → Fourth model
- Ethane ($C_2H_6$) → Third model
- Propane ($C_3H_8$) → First model