QUESTION IMAGE
Question
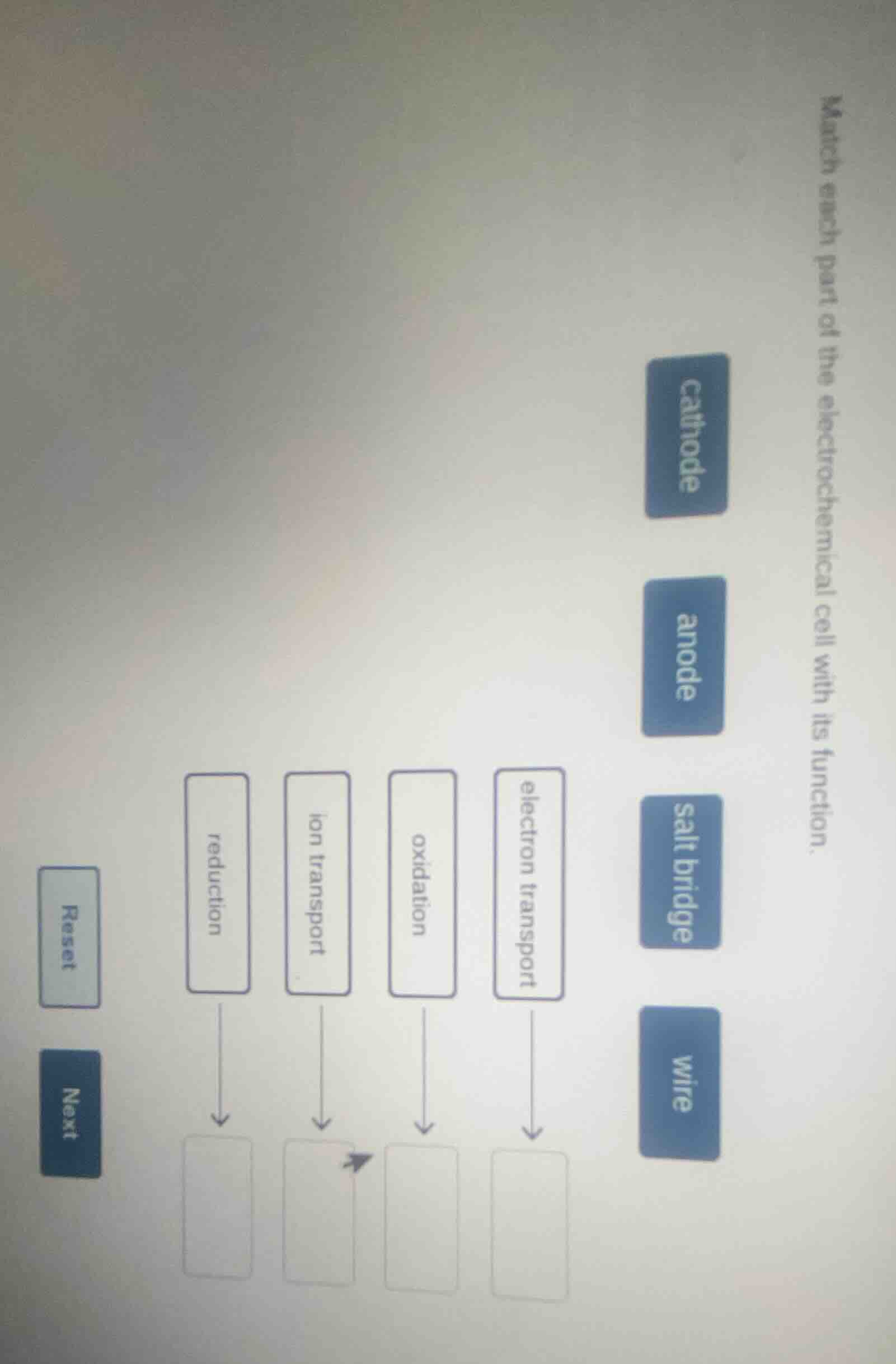
match each part of the electrochemical cell with its function.
reset
next
reduction
ion transport
oxidation
electron transport
cathode
anode
salt bridge
wire
of Electrochemical Cell Parts and Functions:
- Cathode: In an electrochemical cell, the cathode is the site where reduction (gain of electrons) occurs. So, "reduction" matches with "cathode".
- Anode: The anode is the site of oxidation (loss of electrons) in an electrochemical cell. Thus, "oxidation" matches with "anode".
- Salt Bridge: The salt bridge is responsible for the transport of ions between the two half - cells to maintain electrical neutrality. So, "ion transport" matches with "salt bridge".
- Wire: The wire in an electrochemical cell is the path through which electrons move from the anode to the cathode. Hence, "electron transport" matches with "wire".
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Cathode - reduction
- Anode - oxidation
- Salt Bridge - ion transport
- Wire - electron transport