QUESTION IMAGE
Question
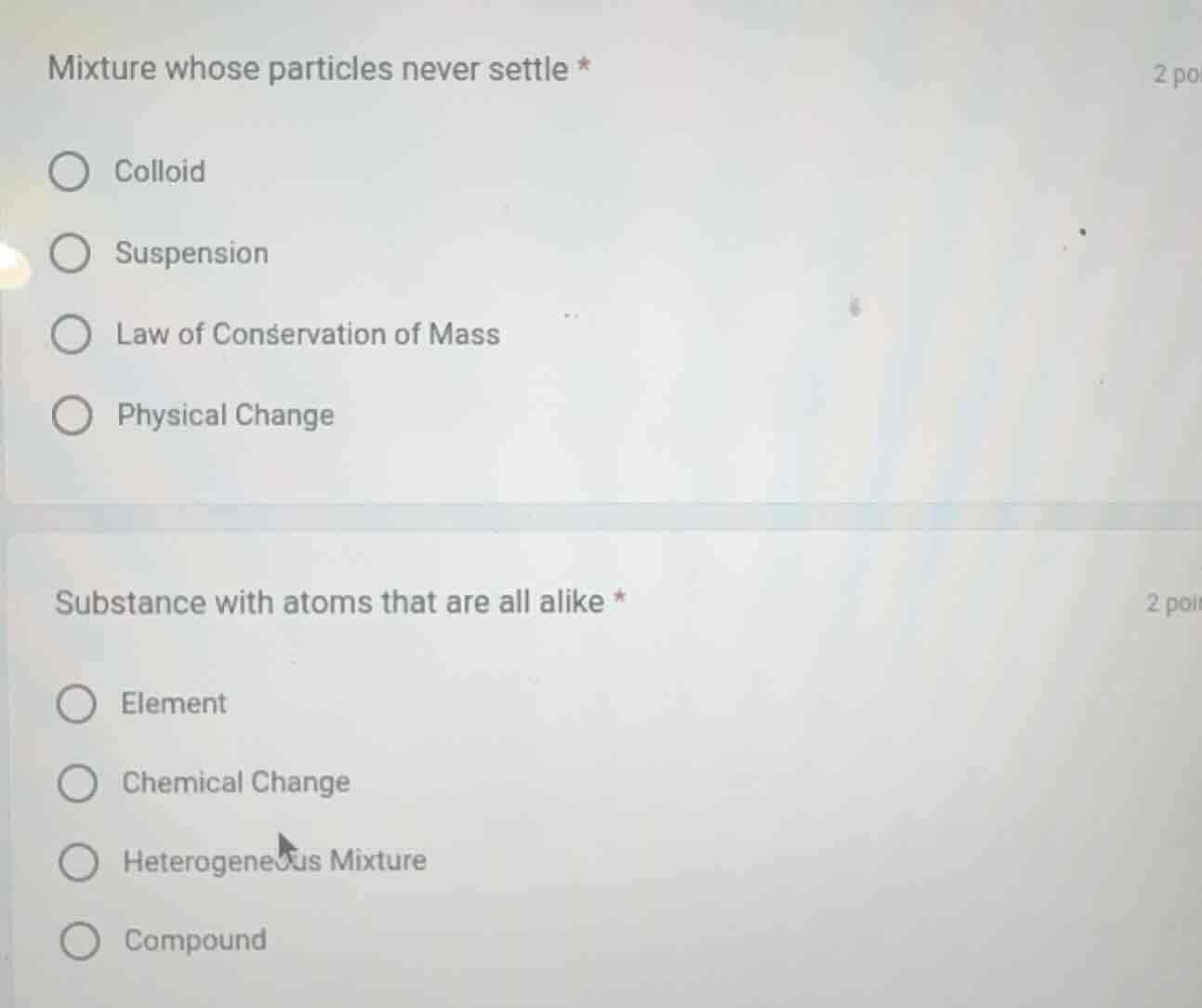
mixture whose particles never settle *
colloid
suspension
law of conservation of mass
physical change
substance with atoms that are all alike *
element
chemical change
heterogeneous mixture
compound
First Question (Mixture whose particles never settle)
A colloid is a mixture where particles are dispersed and do not settle over time. A suspension has particles that settle, the Law of Conservation of Mass is about mass in reactions, and a physical change is about state/ appearance change, not particle settling. So the correct option is Colloid.
An element is a substance made of only one type of atom (all atoms alike). A chemical change involves new substances, a heterogeneous mixture has different components, and a compound has different atoms bonded. So the correct option is Element.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. Colloid