QUESTION IMAGE
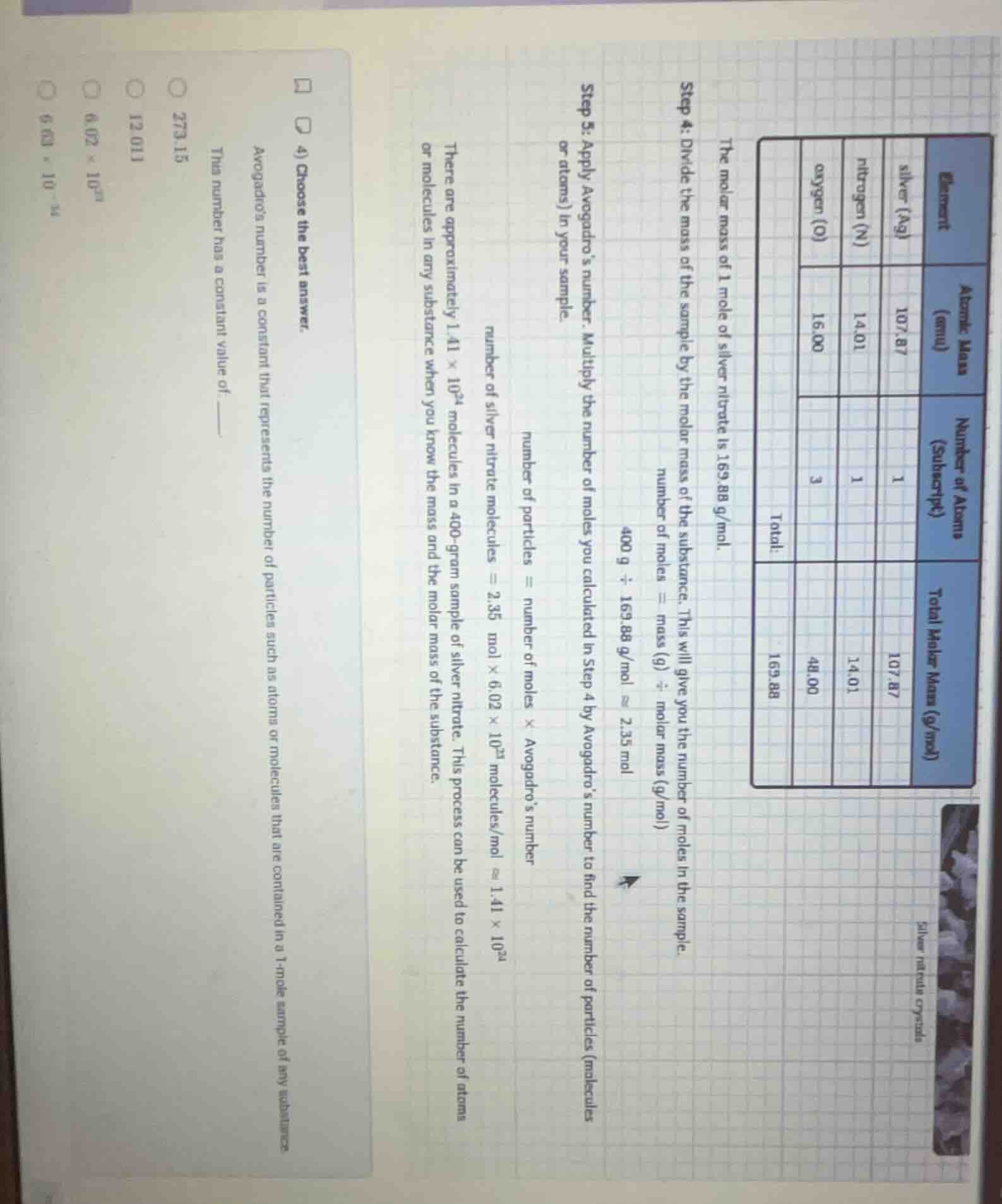
Question
the molar mass of 1 mole of silver nitrate is 169.88 g/mol.
step 4: divide the mass of the sample by the molar mass of the substance. this will give you the number of moles in the sample.
number of moles = mass (g) ÷ molar mass (g/mol)
400 g ÷ 169.88 g/mol ≈ 2.35 mol
step 5: apply avogadro’s number. multiply the number of moles you calculated in step 4 by avogadro’s number to find the number of particles (molecules or atoms) in your sample.
number of particles = number of moles × avogadro’s number
number of silver nitrate molecules = 2.35 mol × 6.02 × 10²³ molecules/mol ≈ 1.41 × 10²⁴
there are approximately 1.41 × 10²⁴ molecules in a 400 - gram sample of silver nitrate. this process can be used to calculate the number of atoms or molecules in any substance when you know the mass and the molar mass of the substance.
- choose the best answer.
avogadro’s number is a constant that represents the number of particles such as atoms or molecules that are contained in a 1 - mole sample of any substance.
this number has a constant value of ______
options:
273.15
12.011
6.02 × 10²³
6.03 × 10⁻²³
Step1: Identify Avogadro's number
Avogadro's number is defined as the number of particles (atoms, molecules, etc.) in 1 mole of a substance, with a constant value of approximately $6.02 \times 10^{23}$.
Step2: Match with options
Compare the definition and value to the given multiple-choice options.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$\boldsymbol{6.02 \times 10^{23}}$ (Option B)