QUESTION IMAGE
Question
multiple choice 1 point
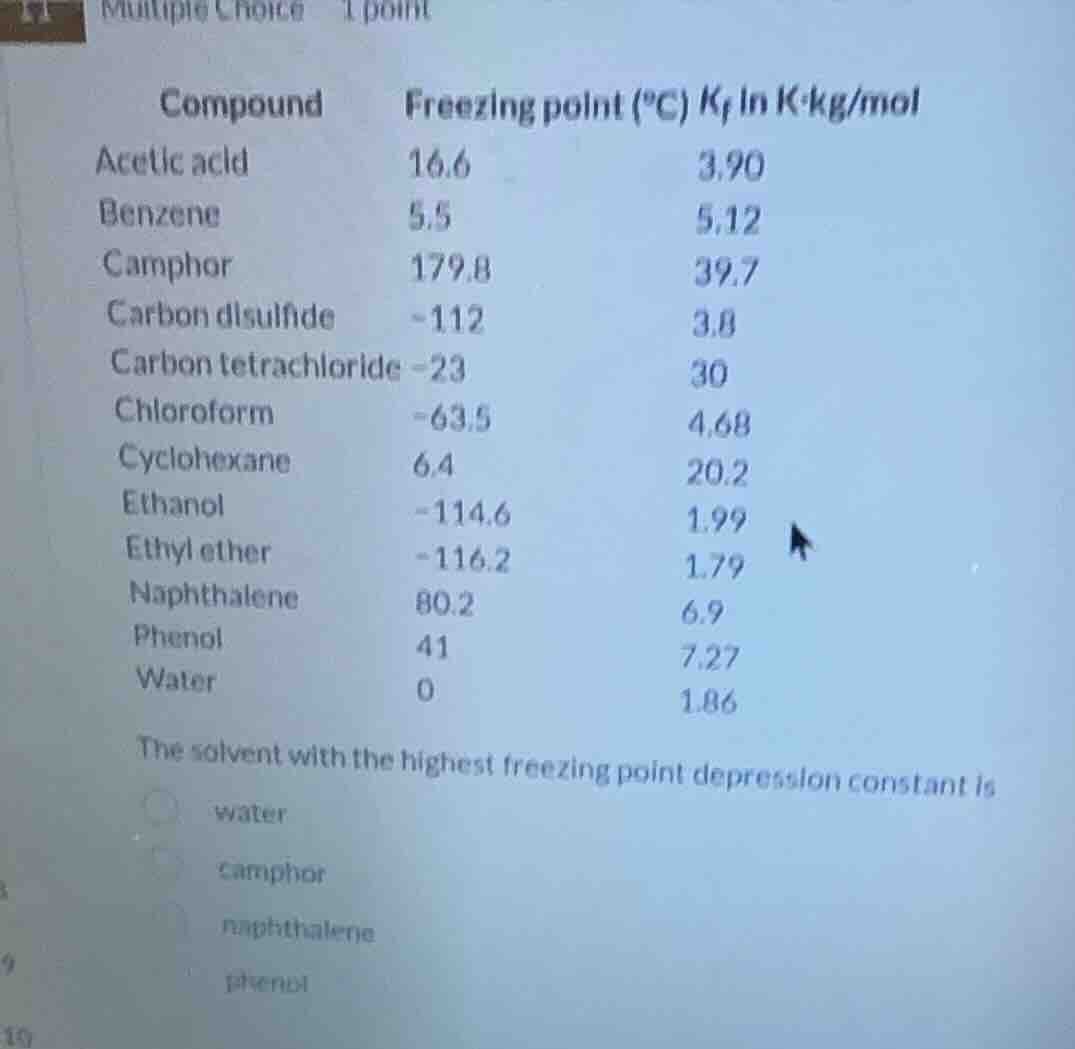
compound freezing point (°c) $k_f$ in k·kg/mol
acetic acid 16.6 3.90
benzene 5.5 5.12
camphor 179.8 39.7
carbon disulfide -112 3.8
carbon tetrachloride -23 30
chloroform -63.5 4.68
cyclohexane 6.4 20.2
ethanol -114.6 1.99
ethyl ether -116.2 1.79
naphthalene 80.2 6.9
phenol 41 7.27
water 0 1.86
the solvent with the highest freezing point depression constant is
water
camphor
naphthalene
phenol
Compare the $K_f$ (freezing point depression constant) values of all listed solvents to find the maximum value. The $K_f$ values are: water ($1.86$), camphor ($39.7$), naphthalene ($6.9$), phenol ($7.27$). Camphor has the highest $K_f$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Camphor