QUESTION IMAGE
Question
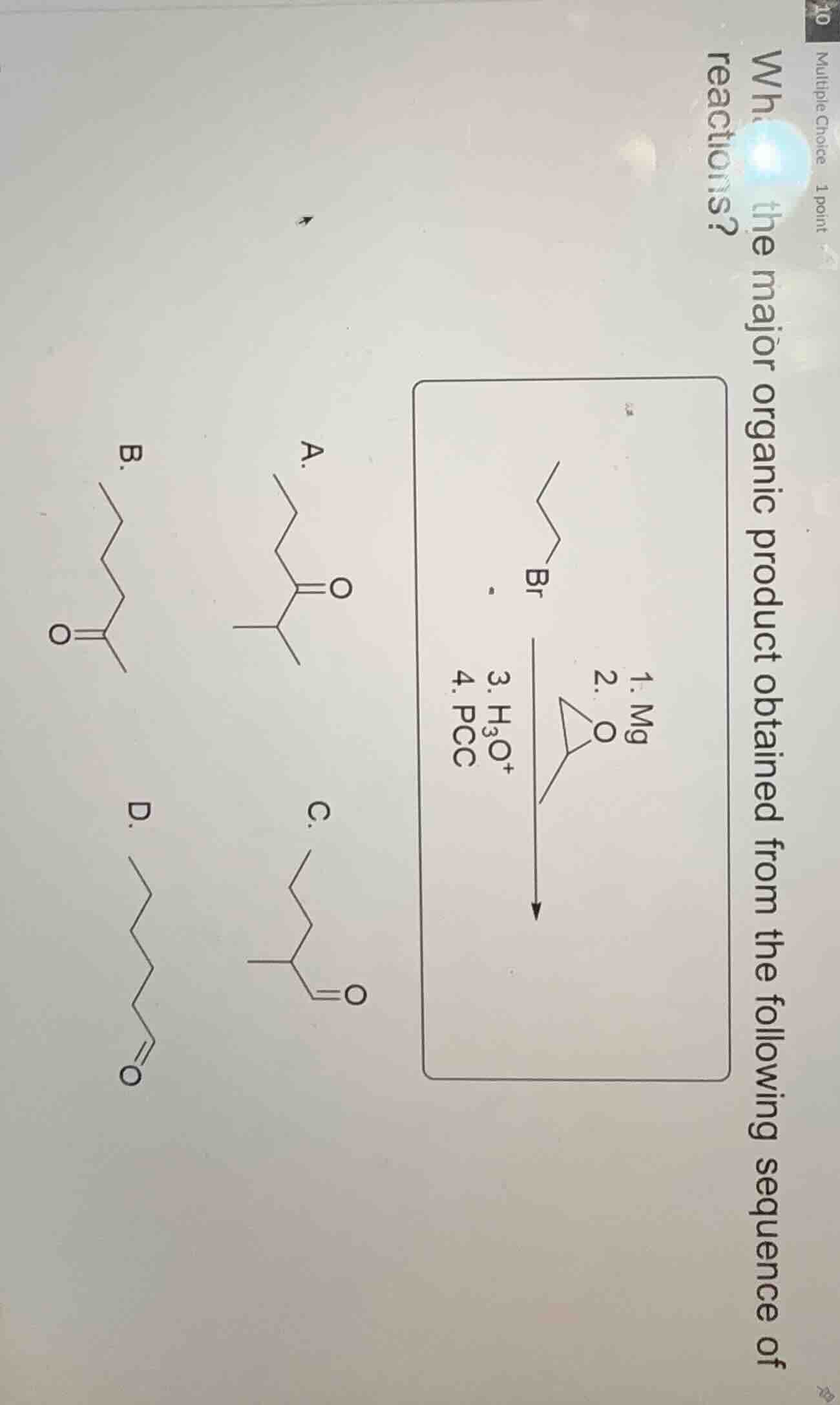
multiple choice 1 point what is the major organic product obtained from the following sequence of reactions?
(reaction sequence and options with chemical structures: starting with a bromo - alkane, then steps 1. mg, 2. (epoxide), 3. h₃o⁺, 4. pcc; options a - d with different carbonyl - containing or other organic structures)
Step1: Analyze the first step (Grignard reagent formation)
The starting material is a bromoalkane. When reacted with Mg, it forms a Grignard reagent (\( \text{R-MgBr} \)), where \( \text{R} \) is the alkyl group from the bromoalkane.
Step2: Reaction with epoxide
Grignard reagents react with epoxides via ring - opening. The Grignard reagent attacks the less substituted carbon of the epoxide (due to steric and electronic factors). The epoxide ring opens, and the alkyl group from the Grignard reagent is attached to the carbon, and the \( \text{-O}^- \) part is protonated later.
Step3: Protonation (work - up with \( \text{H}_3\text{O}^+ \))
After the ring - opening reaction with the Grignard reagent, the alkoxide intermediate formed is protonated by \( \text{H}_3\text{O}^+ \) to form an alcohol.
Step4: Oxidation with PCC
Pyridinium chlorochromate (PCC) is a mild oxidizing agent. It oxidizes primary alcohols to aldehydes and secondary alcohols to ketones. The alcohol formed in the previous step: let's trace the carbon chain. The original bromoalkane has a certain number of carbons, and after adding the carbon from the epoxide ring - opening, we get an alcohol. If the alcohol is a secondary alcohol, PCC will oxidize it to a ketone.
Let's consider the carbon chain: The bromoalkane is propyl bromide (3 - carbon chain). The epoxide is ethylene oxide (2 - carbon ring). After Grignard formation (\( \text{CH}_3\text{CH}_2\text{CH}_2\text{-MgBr} \)), reaction with epoxide (ring - opening) and protonation gives \( \text{CH}_3\text{CH}_2\text{CH}_2\text{CH}_2\text{CH}_2\text{-OH} \)? Wait, no, let's do it more carefully. Wait, the epoxide is a 3 - membered ring? Wait, the epoxide structure: if it's ethylene oxide (oxirane, 2 - carbon ring), the Grignard reagent (propyl magnesium bromide) attacks one of the carbons of the epoxide. The ring opens, and we get a primary alcohol? Wait, no, maybe the epoxide is a substituted epoxide? Wait, looking at the product options, let's re - evaluate.
Wait, the starting bromoalkane: let's count the carbons. The bromoalkane has 3 carbons (propyl bromide: \( \text{CH}_3\text{CH}_2\text{CH}_2\text{Br} \)). The epoxide: if it's a 3 - carbon - related epoxide? Wait, no, the key is the oxidation step. After the Grignard - epoxide - protonation sequence, we have an alcohol. Then PCC oxidation:
If the alcohol is secondary:
Let's consider the structure of the product after protonation. Suppose the alcohol formed is a secondary alcohol. PCC oxidizes secondary alcohols to ketones.
Looking at the options:
- Option A: It's a ketone. Let's check the carbon chain. The bromoalkane (propyl) + epoxide (let's say 2 - carbon ring opening) + oxidation. The carbon chain length and the ketone group: when we have a secondary alcohol (formed from the reaction sequence) and oxidize it with PCC, we get a ketone.
- Option B: It's a ketone but the carbon chain seems different.
- Option C: It's an aldehyde, but PCC on a primary alcohol gives aldehyde, but our reaction sequence (Grignard with epoxide) should give a secondary alcohol (so oxidation to ketone).
- Option D: It's an aldehyde, but again, the alcohol formed should be secondary.
So the major product is the ketone as in option A.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. The ketone - containing compound (the structure with the carbonyl group on the second carbon of the 5 - carbon chain, with a methyl and an ethyl group attached to the carbonyl - adjacent carbon)