QUESTION IMAGE
Question
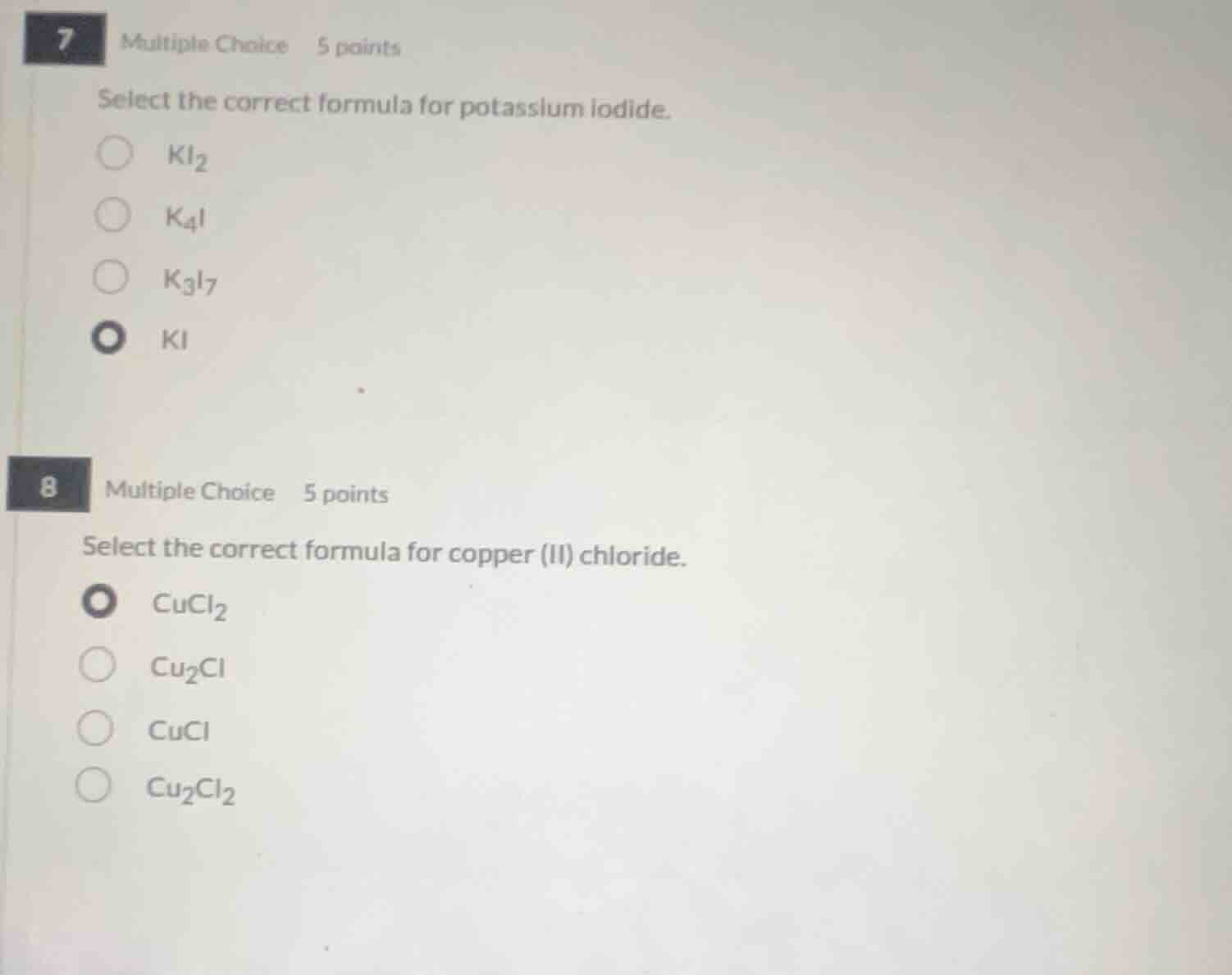
7 multiple choice 5 points
select the correct formula for potassium iodide.
○ ki₂
○ k₄i
○ k₃i₇
● ki
8 multiple choice 5 points
select the correct formula for copper (ii) chloride.
● cucl₂
○ cu₂cl
○ cucl
○ cu₂cl₂
Step1: Analyze potassium iodide ions
Potassium (K) has a +1 charge ($\text{K}^+$), iodide (I) has a -1 charge ($\text{I}^-$). To balance charge, 1 $\text{K}^+$ pairs with 1 $\text{I}^-$.
Step2: Analyze copper (II) chloride ions
Copper (II) has a +2 charge ($\text{Cu}^{2+}$), chloride (Cl) has a -1 charge ($\text{Cl}^-$). To balance charge, 1 $\text{Cu}^{2+}$ pairs with 2 $\text{Cl}^-$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- KI
- $\text{CuCl}_2$