QUESTION IMAGE
Question
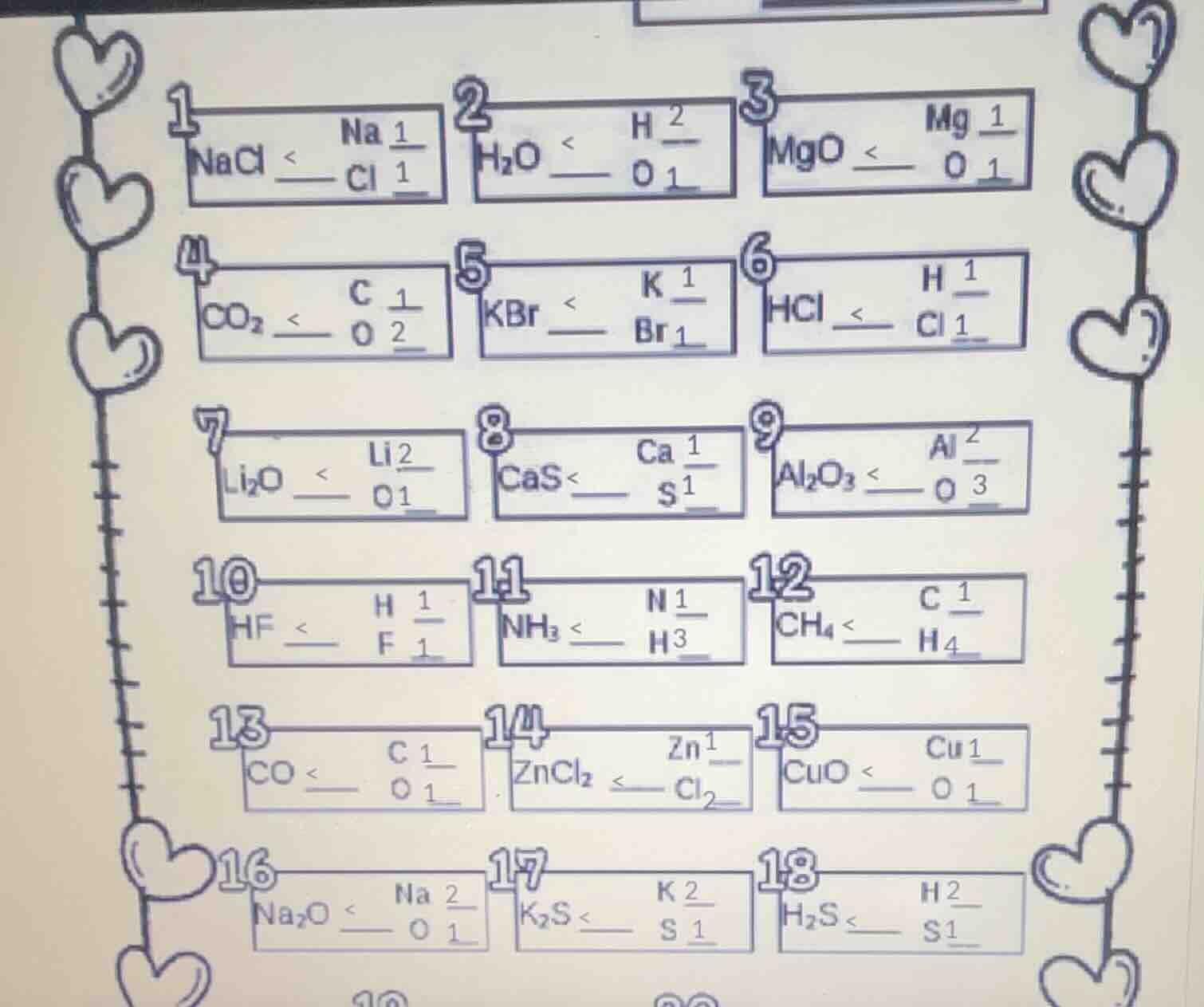
1 nacl na 1/1 cl 1/1
2 h₂o h 2/1 o 1/1
3 mgo mg 1/1 o 1/1
4 co₂ c 1/1 o 2/1
5 kbr k 1/1 br 1/1
6 hcl h 1/1 cl 1/1
7 li₂o li 2/1 o 1/1
8 cas ca 1/1 s 1/1
9 al₂o₃ al 2/1 o 3/1
10 hf h 1/1 f 1/1
11 nh₃ n 1/1 h 3/1
12 ch₄ c 1/1 h 4/1
13 co c 1/1 o 1/1
14 zncl₂ zn 1/1 cl 2/1
15 cuo cu 1/1 o 1/1
16 na₂o na 2/1 o 1/1
17 k₂s k 2/1 s 1/1
18 h₂s h 2/1 s 1/1
To solve this, we need to determine the ratio of the elements in each compound and check if the subscript in the formula matches the ratio. Let's take each compound one by one:
1. NaCl
- Na: 1, Cl: 1. The formula is NaCl (1:1), so it's correct. Mark with
=.
2. H₂O
- H: 2, O: 1. The formula is H₂O (2:1), so it's correct. Mark with
=.
3. MgO
- Mg: 1, O: 1. The formula is MgO (1:1), so it's correct. Mark with
=.
4. CO₂
- C: 1, O: 2. The formula is CO₂ (1:2), so it's correct. Mark with
=.
5. KBr
- K: 1, Br: 1. The formula is KBr (1:1), so it's correct. Mark with
=.
6. HCl
- H: 1, Cl: 1. The formula is HCl (1:1), so it's correct. Mark with
=.
7. Li₂O
- Li: 2, O: 1. The formula is Li₂O (2:1), so it's correct. Mark with
=.
8. CaS
- Ca: 1, S: 1. The formula is CaS (1:1), so it's correct. Mark with
=.
9. Al₂O₃
- Al: 2, O: 3. The formula is Al₂O₃ (2:3), so it's correct. Mark with
=.
10. HF
- H: 1, F: 1. The formula is HF (1:1), so it's correct. Mark with
=.
11. NH₃
- N: 1, H: 3. The formula is NH₃ (1:3), so it's correct. Mark with
=.
12. CH₄
- C: 1, H: 4. The formula is CH₄ (1:4), so it's correct. Mark with
=.
13. CO
- C: 1, O: 1. The formula is CO (1:1), so it's correct. Mark with
=.
14. ZnCl₂
- Zn: 1, Cl: 2. The formula is ZnCl₂ (1:2), so it's correct. Mark with
=.
15. CuO
- Cu: 1, O: 1. The formula is CuO (1:1), so it's correct. Mark with
=.
16. Na₂O
- Na: 2, O: 1. The formula is Na₂O (2:1), so it's correct. Mark with
=.
17. K₂S
- K: 2, S: 1. The formula is K₂S (2:1), so it's correct. Mark with
=.
18. H₂S
- H: 2, S: 1. The formula is H₂S (2:1), so it's correct. Mark with
=.
For each compound (1 - 18), the ratio of elements matches the formula, so we mark each with =.
If we were to write the answers in the boxes (the <___ part), we put = in each box since all ratios match the chemical formulas.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
To solve this, we need to determine the ratio of the elements in each compound and check if the subscript in the formula matches the ratio. Let's take each compound one by one:
1. NaCl
- Na: 1, Cl: 1. The formula is NaCl (1:1), so it's correct. Mark with
=.
2. H₂O
- H: 2, O: 1. The formula is H₂O (2:1), so it's correct. Mark with
=.
3. MgO
- Mg: 1, O: 1. The formula is MgO (1:1), so it's correct. Mark with
=.
4. CO₂
- C: 1, O: 2. The formula is CO₂ (1:2), so it's correct. Mark with
=.
5. KBr
- K: 1, Br: 1. The formula is KBr (1:1), so it's correct. Mark with
=.
6. HCl
- H: 1, Cl: 1. The formula is HCl (1:1), so it's correct. Mark with
=.
7. Li₂O
- Li: 2, O: 1. The formula is Li₂O (2:1), so it's correct. Mark with
=.
8. CaS
- Ca: 1, S: 1. The formula is CaS (1:1), so it's correct. Mark with
=.
9. Al₂O₃
- Al: 2, O: 3. The formula is Al₂O₃ (2:3), so it's correct. Mark with
=.
10. HF
- H: 1, F: 1. The formula is HF (1:1), so it's correct. Mark with
=.
11. NH₃
- N: 1, H: 3. The formula is NH₃ (1:3), so it's correct. Mark with
=.
12. CH₄
- C: 1, H: 4. The formula is CH₄ (1:4), so it's correct. Mark with
=.
13. CO
- C: 1, O: 1. The formula is CO (1:1), so it's correct. Mark with
=.
14. ZnCl₂
- Zn: 1, Cl: 2. The formula is ZnCl₂ (1:2), so it's correct. Mark with
=.
15. CuO
- Cu: 1, O: 1. The formula is CuO (1:1), so it's correct. Mark with
=.
16. Na₂O
- Na: 2, O: 1. The formula is Na₂O (2:1), so it's correct. Mark with
=.
17. K₂S
- K: 2, S: 1. The formula is K₂S (2:1), so it's correct. Mark with
=.
18. H₂S
- H: 2, S: 1. The formula is H₂S (2:1), so it's correct. Mark with
=.
For each compound (1 - 18), the ratio of elements matches the formula, so we mark each with =.
If we were to write the answers in the boxes (the <___ part), we put = in each box since all ratios match the chemical formulas.