QUESTION IMAGE
Question
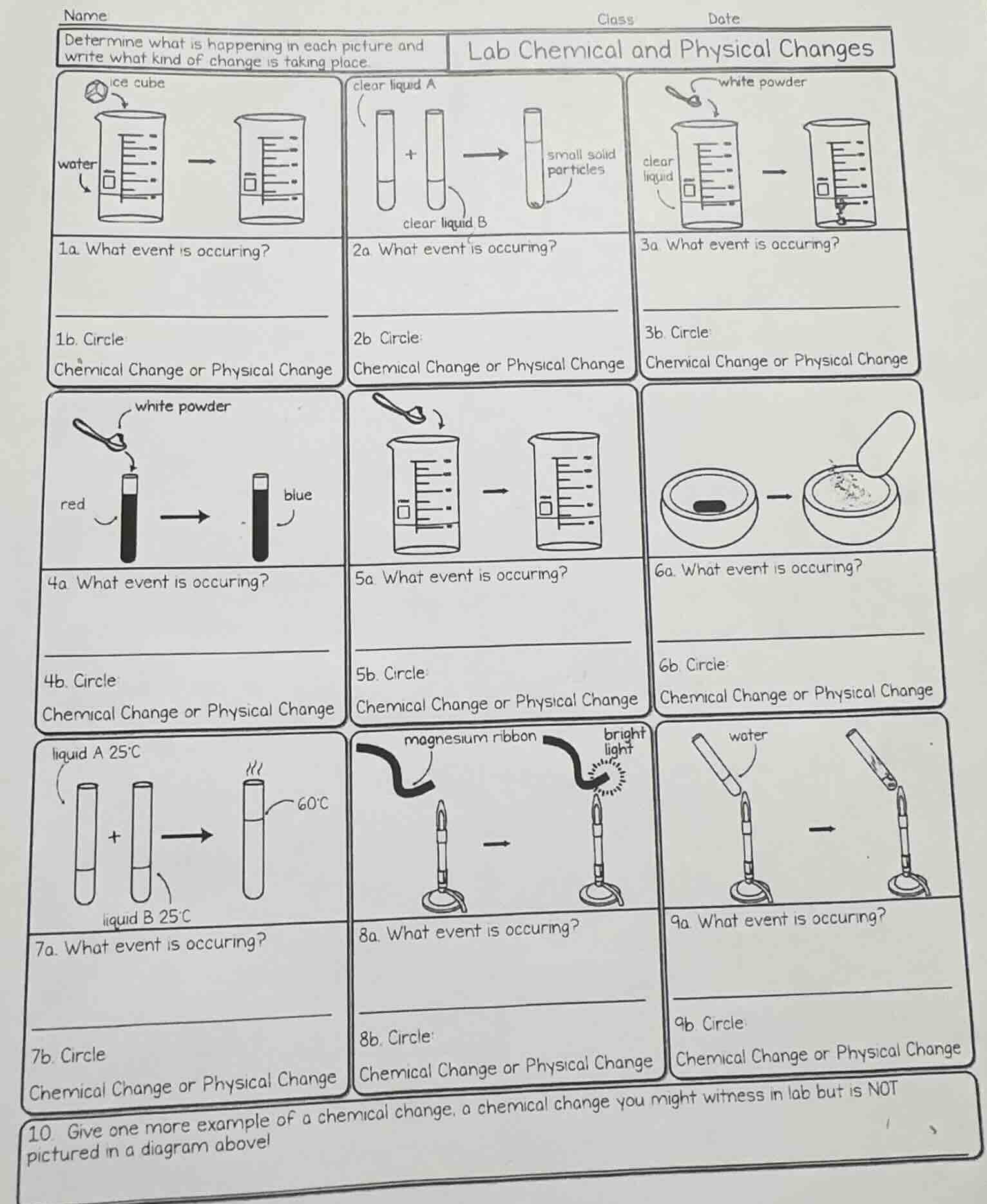
name class date determine what is happening in each picture and write what kind of change is taking place. lab chemical and physical changes 1a. what event is occuring? 1b. circle: chemical change or physical change 2a. what event is occuring? 2b circle: chemical change or physical change 3a. what event is occuring? 3b. circle: chemical change or physical change 4a what event is occuring? 4b. circle: chemical change or physical change 5a. what event is occuring? 5b. circle: chemical change or physical change 6a. what event is occuring? 6b circle: chemical change or physical change 7a. what event is occuring? 7b. circle chemical change or physical change 8a. what event is occuring? 8b. circle: chemical change or physical change 9a. what event is occuring? 9b circle: chemical change or physical change 10 give one more example of a chemical change, a chemical change you might witness in lab but is not pictured in a diagram above!
Step1: Analyze each scenario
1. Ice cube in water
Step1.1: Identify the event
An ice cube melts in water.
Step1.2: Classify the change
Physical Change (no new substance formed; solid water becomes liquid water)
2. Clear liquids A + B form solid
Step2.1: Identify the event
Two clear liquids mix to form a solid precipitate.
Step2.2: Classify the change
Chemical Change (new solid substance is formed)
3. White powder dissolves in liquid
Step3.1: Identify the event
A white powder dissolves in a clear liquid.
Step3.2: Classify the change
Physical Change (the powder disperses, no new substance)
4. White powder changes liquid color
Step4.1: Identify the event
A white powder is added to a red liquid, turning it blue.
Step4.2: Classify the change
Chemical Change (color change indicates new substance formation)
5. White powder dissolves in liquid
Step5.1: Identify the event
A white powder dissolves in a liquid.
Step5.2: Classify the change
Physical Change (no new substance is created)
6. Solid is ground into powder
Step6.1: Identify the event
A solid is crushed into a fine powder with a mortar and pestle.
Step6.2: Classify the change
Physical Change (only particle size changes, no new substance)
7. Liquids A + B heat up
Step7.1: Identify the event
Two liquids mix and the temperature increases.
Step7.2: Classify the change
Chemical Change (temperature change indicates a chemical reaction)
8. Magnesium ribbon burns
Step8.1: Identify the event
A magnesium ribbon burns with a bright light over a flame.
Step8.2: Classify the change
Chemical Change (combustion forms magnesium oxide, a new substance)
9. Water is heated over flame
Step9.1: Identify the event
Water is heated in a test tube over a flame (boiling/heating).
Step9.2: Classify the change
Physical Change (liquid water becomes water vapor, no new substance)
10. Extra chemical change example
Step10.1: Brainstorm a lab example
Iron nails rusting in a beaker of salt water (common lab chemical change not pictured)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
1a. An ice cube melts in water
1b. Physical Change
---
2a. Two clear liquids form a solid precipitate
2b. Chemical Change
---
3a. A white powder dissolves in a clear liquid
3b. Physical Change
---
4a. A white powder changes a red liquid to blue
4b. Chemical Change
---
5a. A white powder dissolves in a liquid
5b. Physical Change
---
6a. A solid is crushed into a fine powder
6b. Physical Change
---
7a. Two liquids mix and increase in temperature
7b. Chemical Change
---
8a. A magnesium ribbon burns with a bright light
8b. Chemical Change
---
9a. Water is heated over a flame (boils)
9b. Physical Change
---
- Rusting of iron nails in salt water (or another valid lab chemical change, e.g., vinegar reacting with baking soda to produce gas bubbles)