QUESTION IMAGE
Question
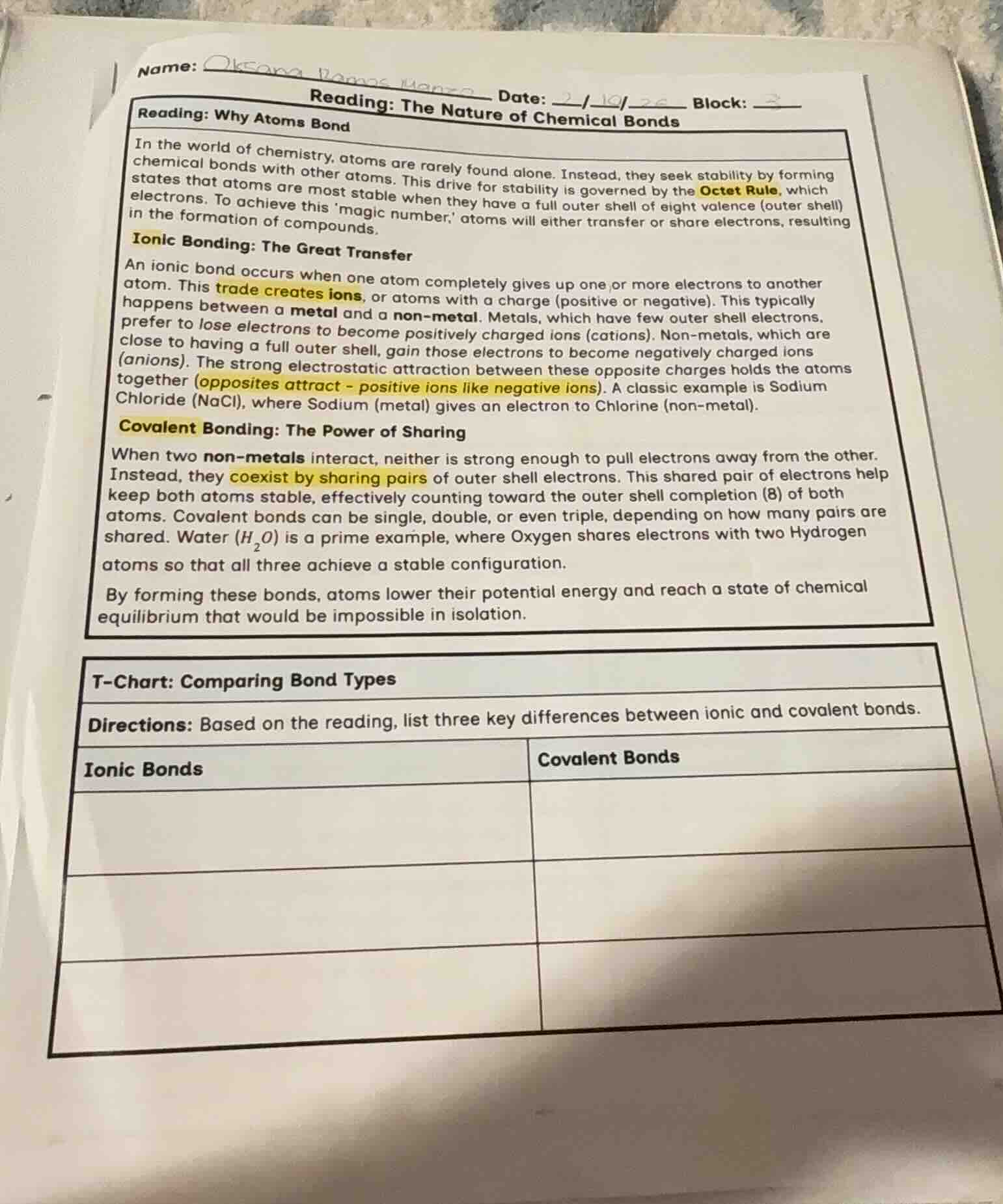
name: date: 2/10/26 block: reading: the nature of chemical bondsreading: why atoms bondin the world of chemistry, atoms are rarely found alone. instead, they seek stability by forming chemical bonds with other atoms. this drive for stability is governed by the octet rule, which states that atoms are most stable when they have a full outer shell of eight valence electrons. to achieve this magic number, atoms will either transfer or share electrons, resulting in the formation of compounds.ionic bonding: the great transferan ionic bond occurs when one atom completely gives up one or more electrons to another atom. this trade creates ions, or atoms with a charge. this typically happens between a metal and a non-metal. metals, which have few outer shell electrons, prefer to lose electrons to become positively charged ions. non-metals, which are close to having a full outer shell, gain those electrons to become negatively charged ions. the strong electrostatic attraction between these opposite charges holds the atoms together. a classic example is sodium chloride, where sodium gives an electron to chlorine.covalent bonding: the power of sharingwhen two non-metals interact, neither is strong enough to pull electrons away from the other. instead, they coexist by sharing pairs of outer shell electrons. this shared pair of electrons help keep both atoms stable, effectively counting toward the outer shell completion of both atoms. covalent bonds can be single, double, or even triple, depending on how many pairs are shared. water ($h_2o$) is a prime example, where oxygen shares electrons with two hydrogen atoms so that all three achieve a stable configuration.by forming these bonds, atoms lower their potential energy and reach a state of chemical equilibrium that would be impossible in isolation.t-chart: comparing bond typesdirections: based on the reading, list three key differences between ionic and covalent bonds.ionic bonds | covalent bonds
The differences are derived directly from the provided reading text, focusing on electron behavior, participating elements, and bond basis.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
| Ionic Bonds | Covalent Bonds |
|---|---|
| Occurs between metal + non-metal | Occurs between two non-metals |
| Held by electrostatic attraction | Held by shared electron pairs |