QUESTION IMAGE
Question
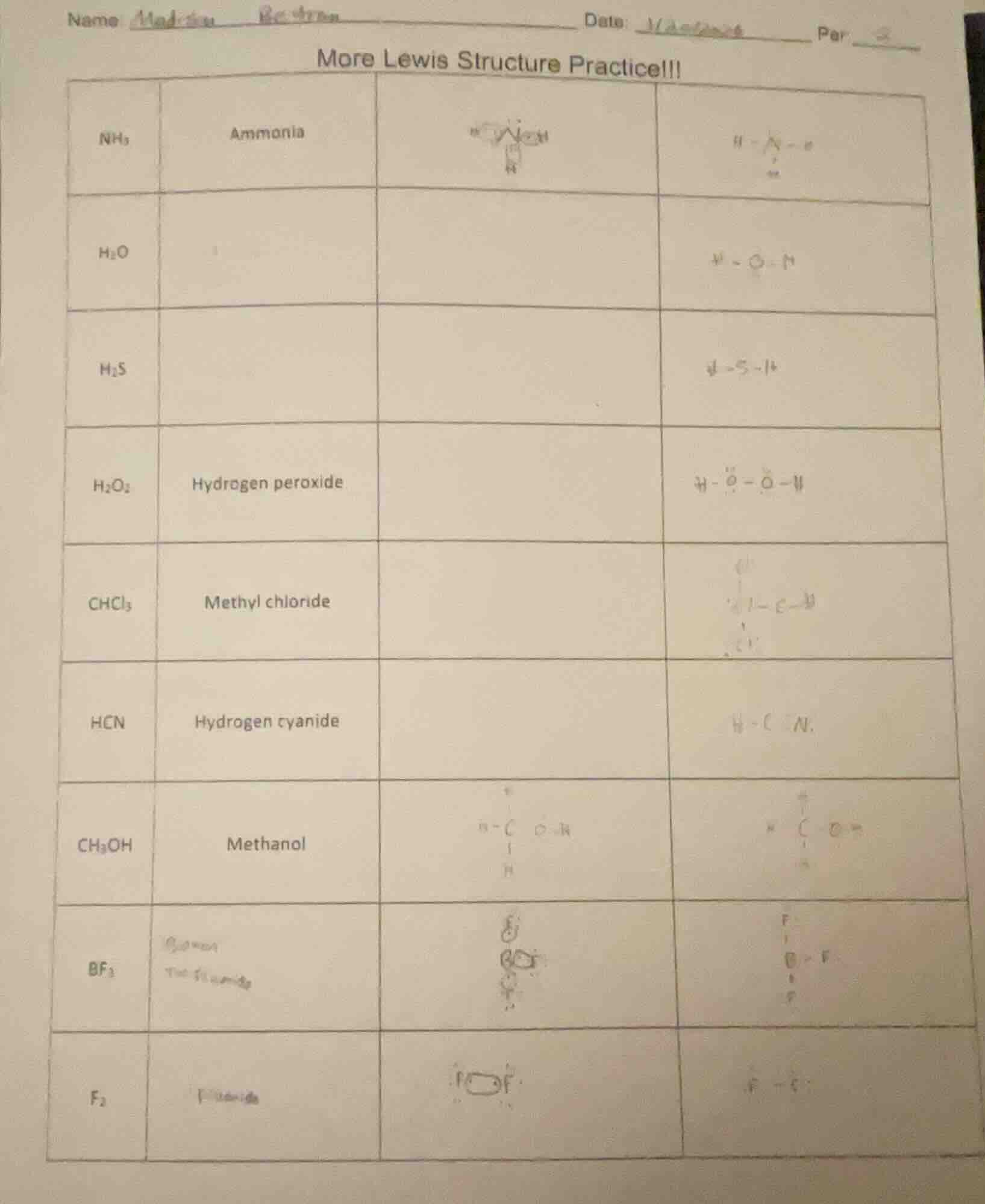
name maddu beckman date: 1/... per __ more lewis structure practice!!! nh₃ ammonia h₂o h₂s h₂o₂ hydrogen peroxide chcl₃ methyl chloride hcn hydrogen cyanide ch₃oh methanol bf₃ f₂
Response
To determine the missing names and complete Lewis structures, we analyze each compound:
1. $\boldsymbol{H_2O}$ (Water)
- Name: Water (common name) or Dihydrogen monoxide (systematic).
- Lewis Structure: Oxygen (O) is central, bonded to 2 H atoms. O has 2 lone pairs. Structure: $ \text{H}-\text{O}-\text{H} $ (with 2 lone pairs on O, often represented as $ \overset{..}{\text{O}} $ between H atoms).
2. $\boldsymbol{H_2S}$ (Hydrogen sulfide)
- Name: Hydrogen sulfide.
- Lewis Structure: Sulfur (S) is central, bonded to 2 H atoms. S has 2 lone pairs. Structure: $ \text{H}-\text{S}-\text{H} $ (with 2 lone pairs on S, similar to $ \text{H}_2\text{O} $ but with S).
3. $\boldsymbol{H_2O_2}$ (Hydrogen peroxide)
- Lewis Structure: Two O atoms bonded (single bond), each bonded to 1 H. Each O has 2 lone pairs. Structure: $ \text{H}-\overset{..}{\text{O}}-\overset{..}{\text{O}}-\text{H} $ (lone pairs on each O).
4. $\boldsymbol{CHCl_3}$ (Chloroform, or Methyl chloride)
- Lewis Structure: Carbon (C) is central, bonded to 1 H and 3 Cl atoms. C has 4 bonds (single bonds to H and each Cl). Structure: $ \text{Cl}-\overset{\text{Cl}}{\underset{\text{Cl}}{\text{C}}}-\text{H} $ (tetrahedral, with Cl atoms around C).
5. $\boldsymbol{HCN}$ (Hydrogen cyanide)
- Lewis Structure: H bonded to C, C triple-bonded to N. Structure: $ \text{H}-\text{C} \equiv \text{N} $ (C has 4 bonds: single to H, triple to N).
6. $\boldsymbol{CH_3OH}$ (Methanol)
- Lewis Structure: C bonded to 3 H and 1 O; O bonded to 1 H. O has 2 lone pairs. Structure: $ \text{H}_3\text{C}-\overset{..}{\text{O}}-\text{H} $ (or $ \text{H}-\overset{\text{H}}{\underset{\text{H}}{\text{C}}}-\overset{..}{\text{O}}-\text{H} $).
7. $\boldsymbol{BF_3}$ (Boron trifluoride)
- Name: Boron trifluoride.
- Lewis Structure: B is central, bonded to 3 F atoms (trigonal planar). B has no lone pairs (incomplete octet). Structure: $ \text{F}-\text{B}-\text{F} $ (with a third F, forming a triangle: $ \overset{\text{F}}{\underset{\text{F}}{\text{B}}}-\text{F} $, each B-F bond is single, and B has 6 valence electrons).
8. $\boldsymbol{F_2}$ (Fluorine gas)
- Name: Fluorine.
- Lewis Structure: Two F atoms bonded (single bond), each with 3 lone pairs. Structure: $ \text{F}-\text{F} $ (with 3 lone pairs on each F: $ \overset{..}{\text{F}}-\overset{..}{\text{F}} $).
Final Table (Completed)
| Formula | Name | Lewis Structure (Simplified) |
|---|---|---|
| $ \text{H}_2\text{O} $ | Water | $ \text{H}-\overset{..}{\text{O}}-\text{H} $ (lone pairs on O) |
| $ \text{H}_2\text{S} $ | Hydrogen sulfide | $ \text{H}-\overset{..}{\text{S}}-\text{H} $ (lone pairs on S) |
| $ \text{H}_2\text{O}_2 $ | Hydrogen peroxide | $ \text{H}-\overset{..}{\text{O}}-\overset{..}{\text{O}}-\text{H} $ |
| $ \text{CHCl}_3 $ | Chloroform (Methyl chloride) | $ \text{Cl}-\overset{\text{Cl}}{\underset{\text{Cl}}{\text{C}}}-\text{H} $ |
| $ \text{HCN} $ | Hydrogen cyanide | $ \text{H}-\text{C} \equiv \text{N} $ |
| $ \text{CH}_3\text{OH} $ | Methanol | $ \text{H}_3\text{C}-\overset{..}{\text{O}}-\text{H} $ |
| $ \text{BF}_3 $ | Boron trifluoride | $ \text{F}-\text{B}-\text{F} $ (trigonal planar, third F omitted for simplicity) |
| $ \text{F}_2 $ | Fluorine | $ \overset{..}{\text{F}}-\overset{..}{\text{F}} $ (3 lone pairs on each F) |
Fo…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
To determine the missing names and complete Lewis structures, we analyze each compound:
1. $\boldsymbol{H_2O}$ (Water)
- Name: Water (common name) or Dihydrogen monoxide (systematic).
- Lewis Structure: Oxygen (O) is central, bonded to 2 H atoms. O has 2 lone pairs. Structure: $ \text{H}-\text{O}-\text{H} $ (with 2 lone pairs on O, often represented as $ \overset{..}{\text{O}} $ between H atoms).
2. $\boldsymbol{H_2S}$ (Hydrogen sulfide)
- Name: Hydrogen sulfide.
- Lewis Structure: Sulfur (S) is central, bonded to 2 H atoms. S has 2 lone pairs. Structure: $ \text{H}-\text{S}-\text{H} $ (with 2 lone pairs on S, similar to $ \text{H}_2\text{O} $ but with S).
3. $\boldsymbol{H_2O_2}$ (Hydrogen peroxide)
- Lewis Structure: Two O atoms bonded (single bond), each bonded to 1 H. Each O has 2 lone pairs. Structure: $ \text{H}-\overset{..}{\text{O}}-\overset{..}{\text{O}}-\text{H} $ (lone pairs on each O).
4. $\boldsymbol{CHCl_3}$ (Chloroform, or Methyl chloride)
- Lewis Structure: Carbon (C) is central, bonded to 1 H and 3 Cl atoms. C has 4 bonds (single bonds to H and each Cl). Structure: $ \text{Cl}-\overset{\text{Cl}}{\underset{\text{Cl}}{\text{C}}}-\text{H} $ (tetrahedral, with Cl atoms around C).
5. $\boldsymbol{HCN}$ (Hydrogen cyanide)
- Lewis Structure: H bonded to C, C triple-bonded to N. Structure: $ \text{H}-\text{C} \equiv \text{N} $ (C has 4 bonds: single to H, triple to N).
6. $\boldsymbol{CH_3OH}$ (Methanol)
- Lewis Structure: C bonded to 3 H and 1 O; O bonded to 1 H. O has 2 lone pairs. Structure: $ \text{H}_3\text{C}-\overset{..}{\text{O}}-\text{H} $ (or $ \text{H}-\overset{\text{H}}{\underset{\text{H}}{\text{C}}}-\overset{..}{\text{O}}-\text{H} $).
7. $\boldsymbol{BF_3}$ (Boron trifluoride)
- Name: Boron trifluoride.
- Lewis Structure: B is central, bonded to 3 F atoms (trigonal planar). B has no lone pairs (incomplete octet). Structure: $ \text{F}-\text{B}-\text{F} $ (with a third F, forming a triangle: $ \overset{\text{F}}{\underset{\text{F}}{\text{B}}}-\text{F} $, each B-F bond is single, and B has 6 valence electrons).
8. $\boldsymbol{F_2}$ (Fluorine gas)
- Name: Fluorine.
- Lewis Structure: Two F atoms bonded (single bond), each with 3 lone pairs. Structure: $ \text{F}-\text{F} $ (with 3 lone pairs on each F: $ \overset{..}{\text{F}}-\overset{..}{\text{F}} $).
Final Table (Completed)
| Formula | Name | Lewis Structure (Simplified) |
|---|---|---|
| $ \text{H}_2\text{O} $ | Water | $ \text{H}-\overset{..}{\text{O}}-\text{H} $ (lone pairs on O) |
| $ \text{H}_2\text{S} $ | Hydrogen sulfide | $ \text{H}-\overset{..}{\text{S}}-\text{H} $ (lone pairs on S) |
| $ \text{H}_2\text{O}_2 $ | Hydrogen peroxide | $ \text{H}-\overset{..}{\text{O}}-\overset{..}{\text{O}}-\text{H} $ |
| $ \text{CHCl}_3 $ | Chloroform (Methyl chloride) | $ \text{Cl}-\overset{\text{Cl}}{\underset{\text{Cl}}{\text{C}}}-\text{H} $ |
| $ \text{HCN} $ | Hydrogen cyanide | $ \text{H}-\text{C} \equiv \text{N} $ |
| $ \text{CH}_3\text{OH} $ | Methanol | $ \text{H}_3\text{C}-\overset{..}{\text{O}}-\text{H} $ |
| $ \text{BF}_3 $ | Boron trifluoride | $ \text{F}-\text{B}-\text{F} $ (trigonal planar, third F omitted for simplicity) |
| $ \text{F}_2 $ | Fluorine | $ \overset{..}{\text{F}}-\overset{..}{\text{F}} $ (3 lone pairs on each F) |
For example, the missing name for $ \text{H}_2\text{O} $ is "Water", and its Lewis structure is $ \text{H}-\overset{..}{\text{O}}-\text{H} $.