QUESTION IMAGE
Question
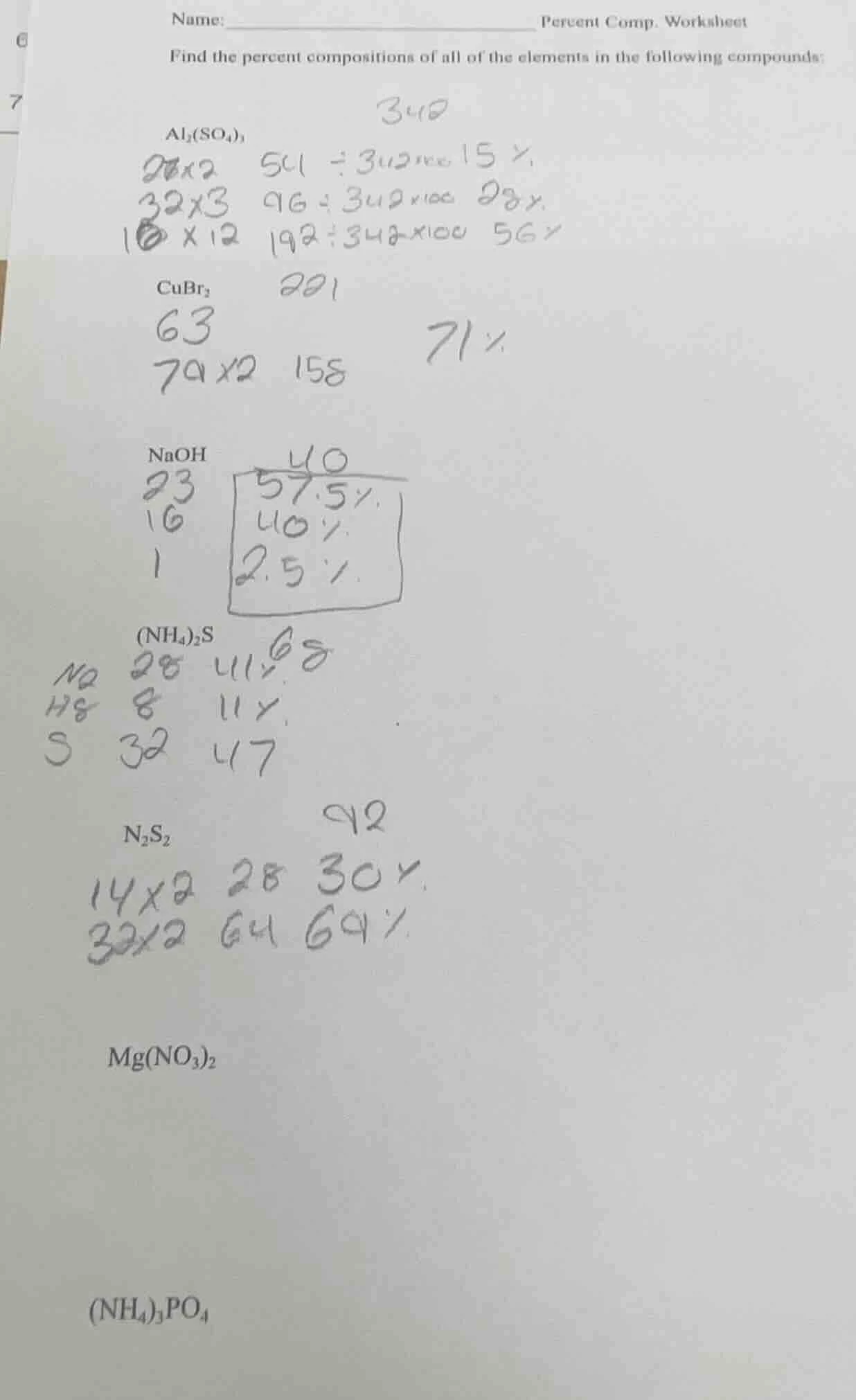
name: _________________________ percent comp. worksheet
find the percent compositions of all of the elements in the following compounds:
$ce{al_{2}(so_{4})_{3}}$
$ce{cubr_{2}}$
$ce{naoh}$
$ce{(nh_{4})_{2}s}$
$ce{n_{2}s_{2}}$
$ce{mg(no_{3})_{2}}$
$ce{(nh_{4})_{3}po_{4}}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$\ce{Al2(SO4)3}$: Al: 15.77%, S: 28.12%, O: 56.11%
$\ce{CuBr2}$: Cu: 28.45%, Br: 71.55%
$\ce{NaOH}$: Na: 57.48%, O: 40.00%, H: 2.52%
$\ce{(NH4)2S}$: N: 41.18%, H: 11.76%, S: 47.06%
$\ce{N2S2}$: N: 30.43%, S: 69.57%
$\ce{Mg(NO3)2}$: Mg: 16.39%, N: 18.97%, O: 64.64%
$\ce{(NH4)3PO4}$: N: 28.17%, H: 8.11%, P: 20.77%, O: 42.95%