QUESTION IMAGE
Question
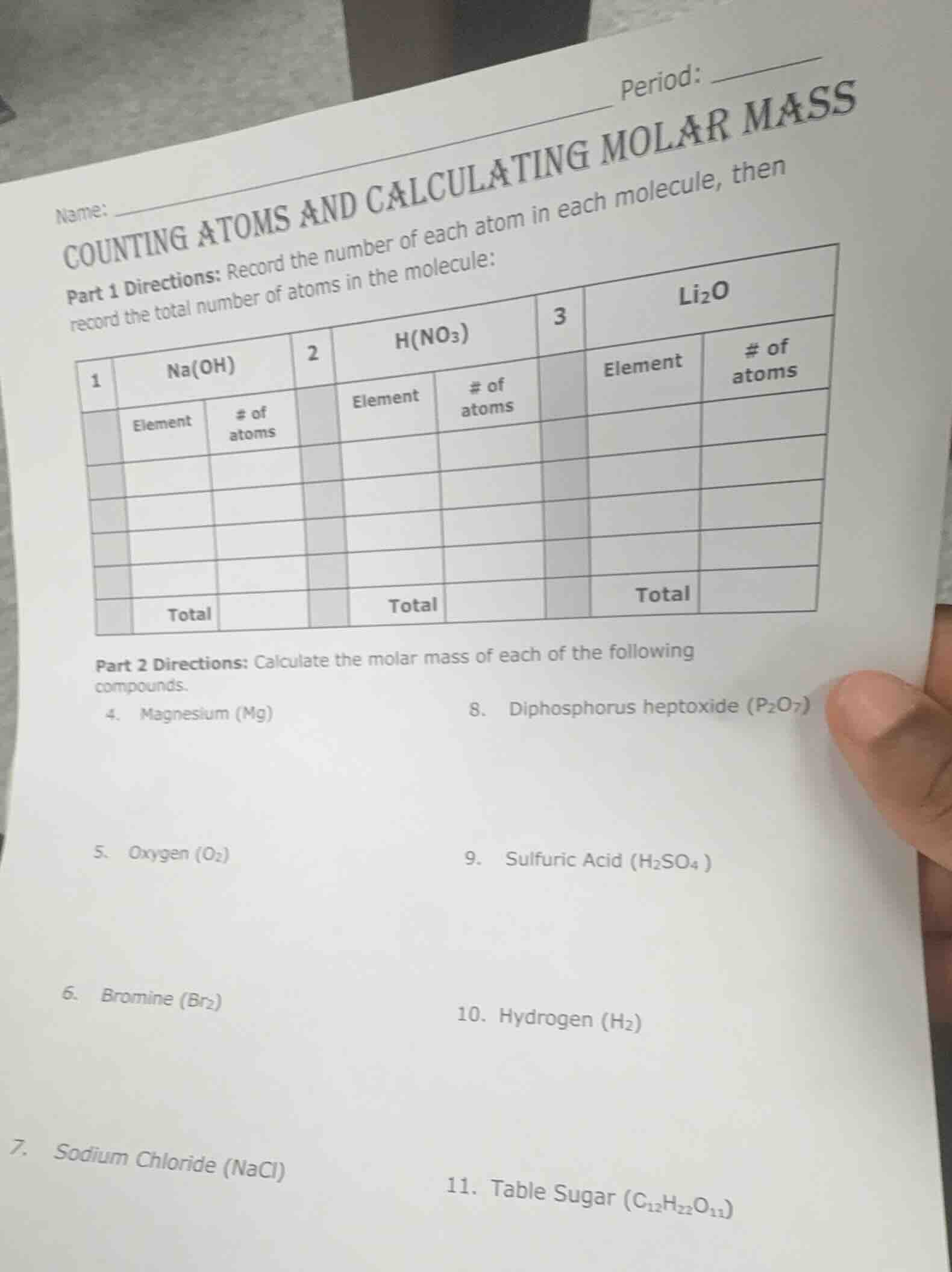
name: ____ period: ____
counting atoms and calculating molar mass
part 1 directions: record the number of each atom in each molecule, then record the total number of atoms in the molecule:
1 na(oh) 2 h(no₃) 3 li₂o
| element | # of atoms | element | # of atoms | element | # of atoms | ||
| total | total | total |
part 2 directions: calculate the molar mass of each of the following compounds.
- magnesium (mg)
- oxygen (o₂)
- bromine (br₂)
- sodium chloride (nacl)
- diphosphorus heptoxide (p₂o₇)
- sulfuric acid (h₂so₄)
- hydrogen (h₂)
- table sugar (c₁₂h₂₂o₁₁)
Part 1: Counting Atoms
For $\boldsymbol{\ce{NaOH}}$:
Step 1: Identify elements and counts
- $\ce{Na}$: 1 atom (no subscript, so 1)
- $\ce{O}$: 1 atom (subscript 1 in $\ce{OH}$)
- $\ce{H}$: 1 atom (subscript 1 in $\ce{OH}$)
Step 2: Calculate total atoms
Total = $1 + 1 + 1 = 3$
For $\boldsymbol{\ce{H(NO3)}}$ (assuming it’s $\ce{HNO3}$):
Step 1: Identify elements and counts
- $\ce{H}$: 1 atom
- $\ce{N}$: 1 atom
- $\ce{O}$: 3 atoms (subscript 3 in $\ce{NO3}$)
Step 2: Calculate total atoms
Total = $1 + 1 + 3 = 5$
For $\boldsymbol{\ce{Li2O}}$:
Step 1: Identify elements and counts
- $\ce{Li}$: 2 atoms (subscript 2)
- $\ce{O}$: 1 atom (no subscript, so 1)
Step 2: Calculate total atoms
Total = $2 + 1 = 3$
Part 2: Calculating Molar Mass (using atomic masses: $\ce{Mg}$=24.31, $\ce{O}$=16.00, $\ce{Br}$=79.90, $\ce{Na}$=22.99, $\ce{Cl}$=35.45, $\ce{P}$=30.97, $\ce{S}$=32.07, $\ce{H}$=1.01, $\ce{C}$=12.01)
4. Magnesium ($\boldsymbol{\ce{Mg}}$):
Molar mass of an element = atomic mass.
Molar mass = $24.31\ \text{g/mol}$
5. Oxygen ($\boldsymbol{\ce{O2}}$):
Step 1: Calculate molar mass of $\ce{O2}$
Molar mass = $2 \times$ atomic mass of $\ce{O}$
= $2 \times 16.00 = 32.00\ \text{g/mol}$
6. Bromine ($\boldsymbol{\ce{Br2}}$):
Step 1: Calculate molar mass of $\ce{Br2}$
Molar mass = $2 \times$ atomic mass of $\ce{Br}$
= $2 \times 79.90 = 159.80\ \text{g/mol}$
7. Sodium Chloride ($\boldsymbol{\ce{NaCl}}$):
Step 1: Sum atomic masses
Molar mass = atomic mass of $\ce{Na}$ + atomic mass of $\ce{Cl}$
= $22.99 + 35.45 = 58.44\ \text{g/mol}$
8. Diphosphorus Heptoxide ($\boldsymbol{\ce{P2O7}}$):
Step 1: Calculate total mass for each element
- $\ce{P}$: $2 \times 30.97 = 61.94$
- $\ce{O}$: $7 \times 16.00 = 112.00$
Step 2: Sum the masses
Molar mass = $61.94 + 112.00 = 173.94\ \text{g/mol}$
9. Sulfuric Acid ($\boldsymbol{\ce{H2SO4}}$):
Step 1: Calculate total mass for each element
- $\ce{H}$: $2 \times 1.01 = 2.02$
- $\ce{S}$: $1 \times 32.07 = 32.07$
- $\ce{O}$: $4 \times 16.00 = 64.00$
Step 2: Sum the masses
Molar mass = $2.02 + 32.07 + 64.00 = 98.09\ \text{g/mol}$
10. Hydrogen ($\boldsymbol{\ce{H2}}$):
Molar mass = $2 \times$ atomic mass of $\ce{H}$
= $2 \times 1.01 = 2.02\ \text{g/mol}$
11. Table Sugar ($\boldsymbol{\ce{C12H22O11}}$):
Step 1: Calculate total mass for each element
- $\ce{C}$: $12 \times 12.01 = 144.12$
- $\ce{H}$: $22 \times 1.01 = 22.22$
- $\ce{O}$: $11 \times 16.00 = 176.00$
Step 2: Sum the masses
Molar mass = $144.12 + 22.22 + 176.00 = 342.34\ \text{g/mol}$
Summary Tables (Part 1)
| Compound | Element | # of Atoms | Element | # of Atoms | Element | # of Atoms | Total Atoms |
|---|---|---|---|---|---|---|---|
| $\ce{HNO3}$ | $\ce{H}$ | 1 | $\ce{N}$ | 1 | $\ce{O}$ | 3 | 5 |
| $\ce{Li2O}$ | $\ce{Li}$ | 2 | $\ce{O}$ | 1 | - | - | 3 |
Final Answers (Molar Masses)
- $\ce{Mg}$: $\boldsymbol{24.31\ \text{g/mol}}$
- $\ce{O2}$: $\boldsymbol{32.00\ \text{g/mol}}$
- $\ce{Br2}$: $\boldsymbol{159.80\ \text{g/mol}}$
- $\ce{NaCl}$: $\boldsymbol{58.44\ \text{g/mol}}$
- $\ce{P2O7}$: $\boldsymbol{173.94\ \text{g/mol}}$
- $\ce{H2SO4}$: $\boldsymbol{98.09\ \text{g/mol}}$
- $\ce{H2}$: $\boldsymbol{2.02\ \text{g/mol}}$
- $\ce{C12H22O11}$: $\boldsymbol{342.34\ \text{g/mol}}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Part 1: Counting Atoms
For $\boldsymbol{\ce{NaOH}}$:
Step 1: Identify elements and counts
- $\ce{Na}$: 1 atom (no subscript, so 1)
- $\ce{O}$: 1 atom (subscript 1 in $\ce{OH}$)
- $\ce{H}$: 1 atom (subscript 1 in $\ce{OH}$)
Step 2: Calculate total atoms
Total = $1 + 1 + 1 = 3$
For $\boldsymbol{\ce{H(NO3)}}$ (assuming it’s $\ce{HNO3}$):
Step 1: Identify elements and counts
- $\ce{H}$: 1 atom
- $\ce{N}$: 1 atom
- $\ce{O}$: 3 atoms (subscript 3 in $\ce{NO3}$)
Step 2: Calculate total atoms
Total = $1 + 1 + 3 = 5$
For $\boldsymbol{\ce{Li2O}}$:
Step 1: Identify elements and counts
- $\ce{Li}$: 2 atoms (subscript 2)
- $\ce{O}$: 1 atom (no subscript, so 1)
Step 2: Calculate total atoms
Total = $2 + 1 = 3$
Part 2: Calculating Molar Mass (using atomic masses: $\ce{Mg}$=24.31, $\ce{O}$=16.00, $\ce{Br}$=79.90, $\ce{Na}$=22.99, $\ce{Cl}$=35.45, $\ce{P}$=30.97, $\ce{S}$=32.07, $\ce{H}$=1.01, $\ce{C}$=12.01)
4. Magnesium ($\boldsymbol{\ce{Mg}}$):
Molar mass of an element = atomic mass.
Molar mass = $24.31\ \text{g/mol}$
5. Oxygen ($\boldsymbol{\ce{O2}}$):
Step 1: Calculate molar mass of $\ce{O2}$
Molar mass = $2 \times$ atomic mass of $\ce{O}$
= $2 \times 16.00 = 32.00\ \text{g/mol}$
6. Bromine ($\boldsymbol{\ce{Br2}}$):
Step 1: Calculate molar mass of $\ce{Br2}$
Molar mass = $2 \times$ atomic mass of $\ce{Br}$
= $2 \times 79.90 = 159.80\ \text{g/mol}$
7. Sodium Chloride ($\boldsymbol{\ce{NaCl}}$):
Step 1: Sum atomic masses
Molar mass = atomic mass of $\ce{Na}$ + atomic mass of $\ce{Cl}$
= $22.99 + 35.45 = 58.44\ \text{g/mol}$
8. Diphosphorus Heptoxide ($\boldsymbol{\ce{P2O7}}$):
Step 1: Calculate total mass for each element
- $\ce{P}$: $2 \times 30.97 = 61.94$
- $\ce{O}$: $7 \times 16.00 = 112.00$
Step 2: Sum the masses
Molar mass = $61.94 + 112.00 = 173.94\ \text{g/mol}$
9. Sulfuric Acid ($\boldsymbol{\ce{H2SO4}}$):
Step 1: Calculate total mass for each element
- $\ce{H}$: $2 \times 1.01 = 2.02$
- $\ce{S}$: $1 \times 32.07 = 32.07$
- $\ce{O}$: $4 \times 16.00 = 64.00$
Step 2: Sum the masses
Molar mass = $2.02 + 32.07 + 64.00 = 98.09\ \text{g/mol}$
10. Hydrogen ($\boldsymbol{\ce{H2}}$):
Molar mass = $2 \times$ atomic mass of $\ce{H}$
= $2 \times 1.01 = 2.02\ \text{g/mol}$
11. Table Sugar ($\boldsymbol{\ce{C12H22O11}}$):
Step 1: Calculate total mass for each element
- $\ce{C}$: $12 \times 12.01 = 144.12$
- $\ce{H}$: $22 \times 1.01 = 22.22$
- $\ce{O}$: $11 \times 16.00 = 176.00$
Step 2: Sum the masses
Molar mass = $144.12 + 22.22 + 176.00 = 342.34\ \text{g/mol}$
Summary Tables (Part 1)
| Compound | Element | # of Atoms | Element | # of Atoms | Element | # of Atoms | Total Atoms |
|---|---|---|---|---|---|---|---|
| $\ce{HNO3}$ | $\ce{H}$ | 1 | $\ce{N}$ | 1 | $\ce{O}$ | 3 | 5 |
| $\ce{Li2O}$ | $\ce{Li}$ | 2 | $\ce{O}$ | 1 | - | - | 3 |
Final Answers (Molar Masses)
- $\ce{Mg}$: $\boldsymbol{24.31\ \text{g/mol}}$
- $\ce{O2}$: $\boldsymbol{32.00\ \text{g/mol}}$
- $\ce{Br2}$: $\boldsymbol{159.80\ \text{g/mol}}$
- $\ce{NaCl}$: $\boldsymbol{58.44\ \text{g/mol}}$
- $\ce{P2O7}$: $\boldsymbol{173.94\ \text{g/mol}}$
- $\ce{H2SO4}$: $\boldsymbol{98.09\ \text{g/mol}}$
- $\ce{H2}$: $\boldsymbol{2.02\ \text{g/mol}}$
- $\ce{C12H22O11}$: $\boldsymbol{342.34\ \text{g/mol}}$