QUESTION IMAGE
Question
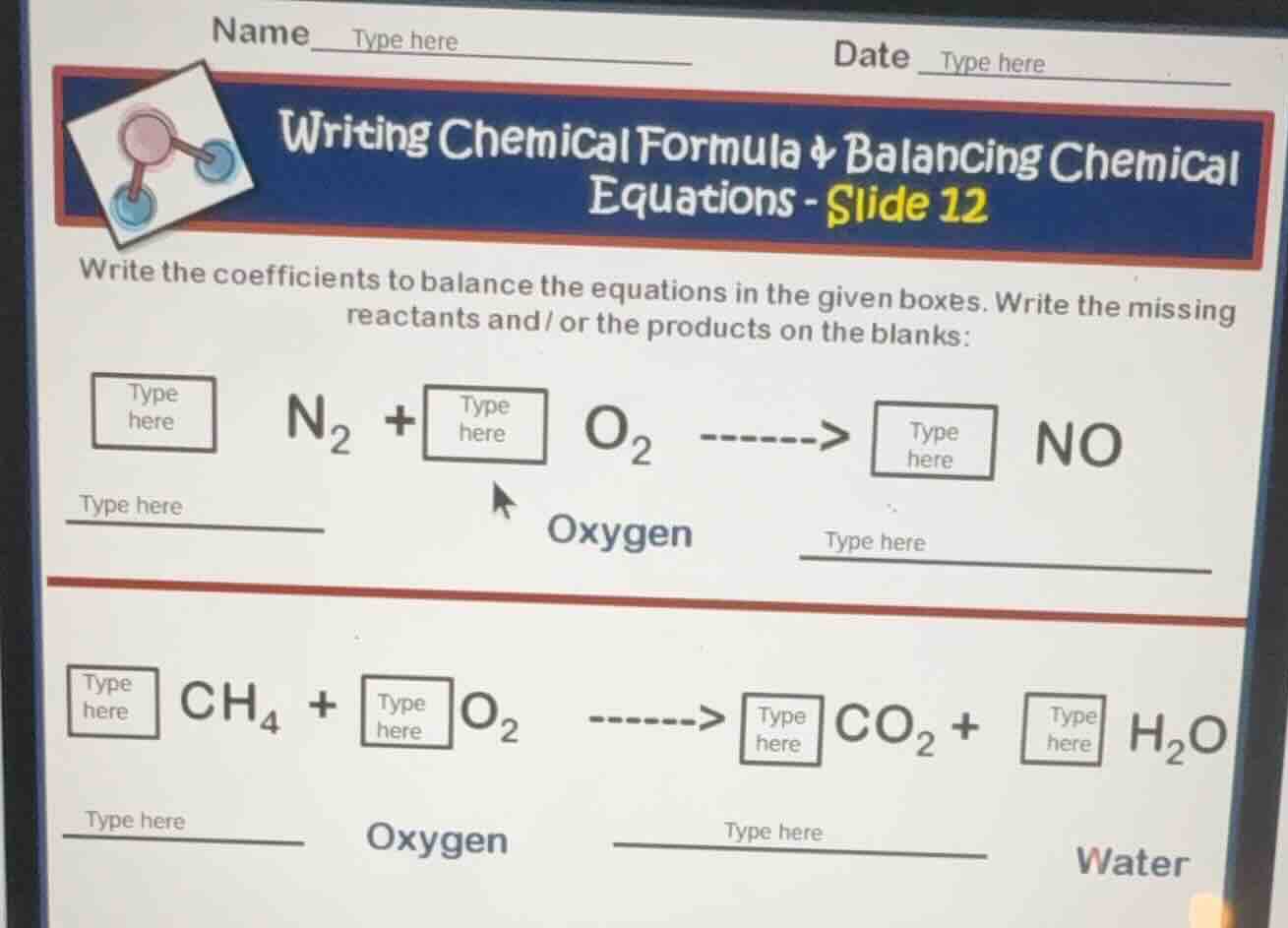
name type here date type here
writing chemical formula & balancing chemical equations - slide 12
write the coefficients to balance the equations in the given boxes. write the missing reactants and/ or the products on the blanks:
$square$ $\text{n}_2$ + $square$ $\text{o}_2$ -------> $square$ $\text{no}$
type here oxygen type here
$square$ $\text{ch}_4$ + $square$ $\text{o}_2$ -------> $square$ $\text{co}_2$ + $square$ $\text{h}_2\text{o}$
type here oxygen type here water
Step1: Balance N in first equation
Count N atoms: left has 2, right has 1. Multiply NO by 2:
$\text{N}_2 + \text{O}_2
ightarrow 2\text{NO}$
Step2: Balance O in first equation
Now O atoms: right has 2, left has 2. Add coefficients:
$1\text{N}_2 + 1\text{O}_2
ightarrow 2\text{NO}$
Step3: Balance C in second equation
C atoms: left has 1, right has 1. $\text{CH}_4 + \text{O}_2
ightarrow 1\text{CO}_2 + \text{H}_2\text{O}$
Step4: Balance H in second equation
H atoms: left has 4, right has 2. Multiply $\text{H}_2\text{O}$ by 2:
$\text{CH}_4 + \text{O}_2
ightarrow \text{CO}_2 + 2\text{H}_2\text{O}$
Step5: Balance O in second equation
O atoms: right has $2 + 2 = 4$, left has 2. Multiply $\text{O}_2$ by 2:
$1\text{CH}_4 + 2\text{O}_2
ightarrow 1\text{CO}_2 + 2\text{H}_2\text{O}$
Step6: Fill missing names
Identify reactants/products: $\text{N}_2$ is Nitrogen, $\text{NO}$ is Nitrogen monoxide; $\text{CH}_4$ is Methane, $\text{CO}_2$ is Carbon dioxide.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
First Equation:
Coefficients: 1 (for $\text{N}_2$), 1 (for $\text{O}_2$), 2 (for $\text{NO}$)
Missing names: Nitrogen (under $\text{N}_2$), Nitrogen monoxide (under $\text{NO}$)
Balanced equation: $\text{N}_2 + \text{O}_2
ightarrow 2\text{NO}$
Second Equation:
Coefficients: 1 (for $\text{CH}_4$), 2 (for $\text{O}_2$), 1 (for $\text{CO}_2$), 2 (for $\text{H}_2\text{O}$)
Missing names: Methane (under $\text{CH}_4$), Carbon dioxide (under $\text{CO}_2$)
Balanced equation: $\text{CH}_4 + 2\text{O}_2
ightarrow \text{CO}_2 + 2\text{H}_2\text{O}$