QUESTION IMAGE
Question
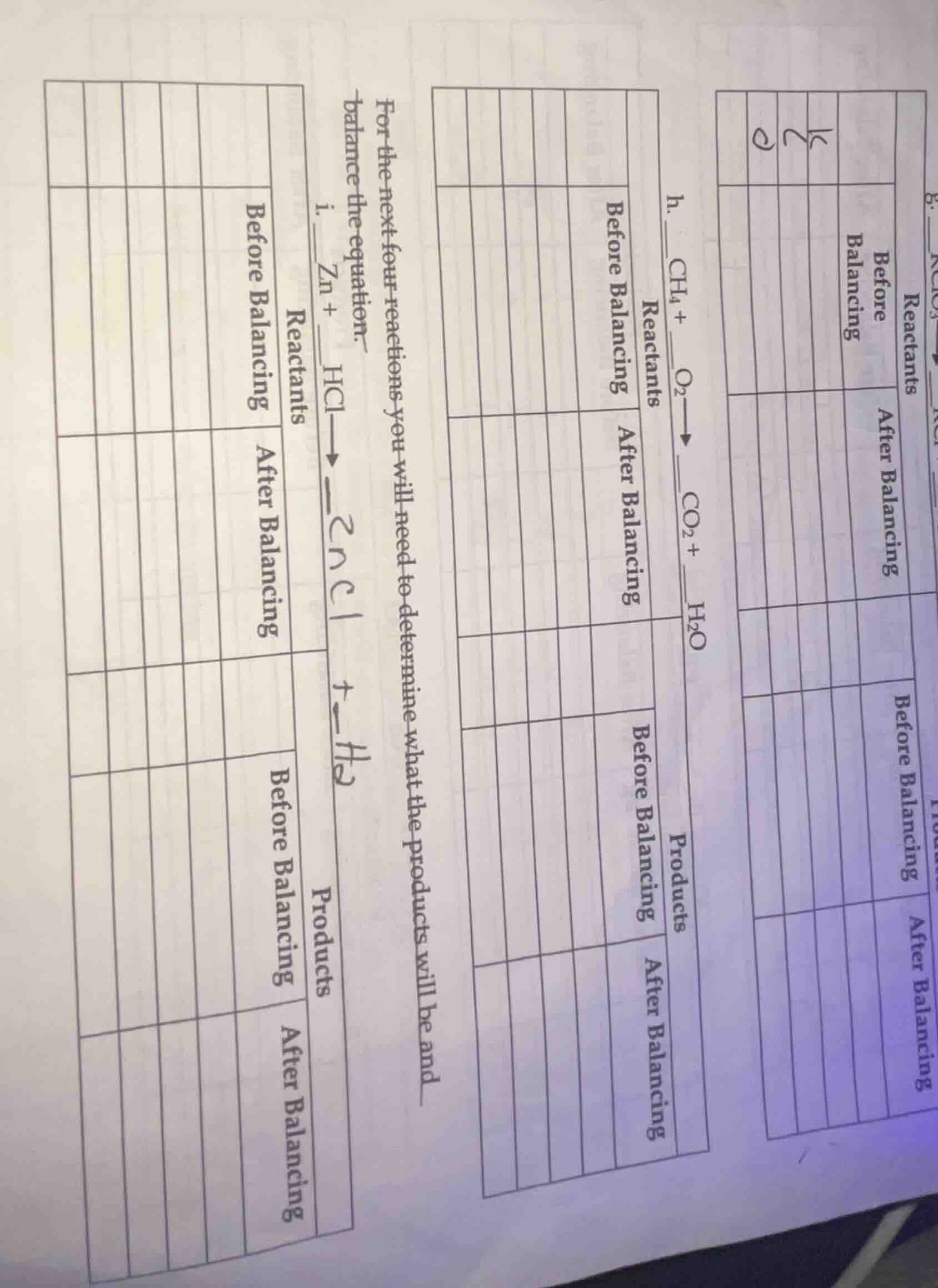
for the next four reactions you will need to determine what the products will be and balance the equation.
i. zn + hcl \
ightarrow zncl + h_2
h. ch_4 + o_2 \
ightarrow co_2 + h_2o
Part g: $\boldsymbol{KClO_3
ightarrow KCl + O_2}$
Step1: Count atoms before balancing
Reactants: $1$ K, $1$ Cl, $3$ O
Products: $1$ K, $1$ Cl, $2$ O
Step2: Balance oxygen atoms
Add coefficient 2 to $\text{KClO}_3$, 2 to $\text{KCl}$, 3 to $\text{O}_2$:
Step3: Verify balanced atoms
Reactants: $2$ K, $2$ Cl, $6$ O
Products: $2$ K, $2$ Cl, $6$ O
---
Part h: $\boldsymbol{CH_4 + O_2
ightarrow CO_2 + H_2O}$
Step1: Count atoms before balancing
Reactants: $1$ C, $4$ H, $2$ O
Products: $1$ C, $2$ H, $3$ O
Step2: Balance hydrogen atoms
Add coefficient 2 to $\text{H}_2\text{O}$:
Step3: Balance oxygen atoms
Add coefficient 2 to $\text{O}_2$:
Step4: Verify balanced atoms
Reactants: $1$ C, $4$ H, $4$ O
Products: $1$ C, $4$ H, $4$ O
---
Part i: $\boldsymbol{Zn + HCl
ightarrow ZnCl_2 + H_2}$
Step1: Count atoms before balancing
Reactants: $1$ Zn, $1$ H, $1$ Cl
Products: $1$ Zn, $2$ H, $2$ Cl
Step2: Balance Cl and H atoms
Add coefficient 2 to $\text{HCl}$:
Step3: Verify balanced atoms
Reactants: $1$ Zn, $2$ H, $2$ Cl
Products: $1$ Zn, $2$ H, $2$ Cl
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Part g:
| Reactants | Before Balancing | After Balancing | Products | Before Balancing | After Balancing |
|---|---|---|---|---|---|
| $\text{O}_2$ | 1 | 3 |
Balanced equation: $2\text{KClO}_3
ightarrow 2\text{KCl} + 3\text{O}_2$
Part h:
| Reactants | Before Balancing | After Balancing | Products | Before Balancing | After Balancing |
|---|---|---|---|---|---|
| $\text{O}_2$ | 1 | 2 | $\text{H}_2\text{O}$ | 1 | 2 |
Balanced equation: $\text{CH}_4 + 2\text{O}_2
ightarrow \text{CO}_2 + 2\text{H}_2\text{O}$
Part i:
| Reactants | Before Balancing | After Balancing | Products | Before Balancing | After Balancing |
|---|---|---|---|---|---|
| $\text{HCl}$ | 1 | 2 | $\text{H}_2$ | 1 | 1 |
Balanced equation: $\text{Zn} + 2\text{HCl}
ightarrow \text{ZnCl}_2 + \text{H}_2$