QUESTION IMAGE
Question
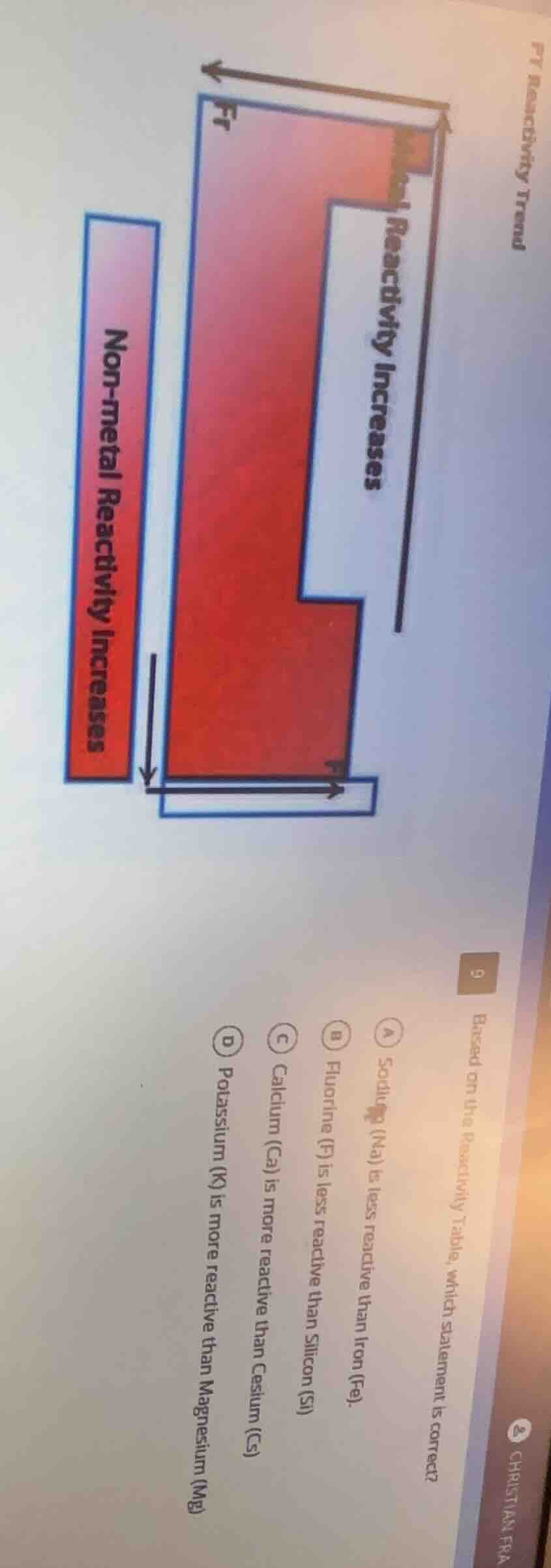
p7 reactivity trend
9
b. based on the reactivity table, which statement is correct?
a sodium (na) is less reactive than iron (fe).
b fluorine (f) is less reactive than silicon (si)
c calcium (ca) is more reactive than cesium (cs)
d potassium (k) is more reactive than magnesium (mg)
To solve this, we analyze each option using the reactivity series (metals: reactivity increases down a group and left along a period; non - metals: reactivity increases up a group and right along a period):
- Option A: In the metal reactivity series, Sodium (Na) is more reactive than Iron (Fe). So this statement is wrong.
- Option B: Fluorine (F) is a non - metal, and Silicon (Si) is a metalloid. Non - metals like F are more reactive than metalloids like Si (as non - metal reactivity increases towards the top right of the periodic table). So this statement is wrong.
- Option C: In the alkali metals/alkaline earth metals series, Cesium (Cs) is more reactive than Calcium (Ca) (reactivity increases down a group). So this statement is wrong.
- Option D: Potassium (K) is in group 1, and Magnesium (Mg) is in group 2. For metals, reactivity increases down a group and left along a period. K is more to the left and lower in the group compared to Mg - related positions, so K is more reactive than Mg. This statement is correct.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
D. Potassium (K) is more reactive than Magnesium (Mg)