QUESTION IMAGE
Question
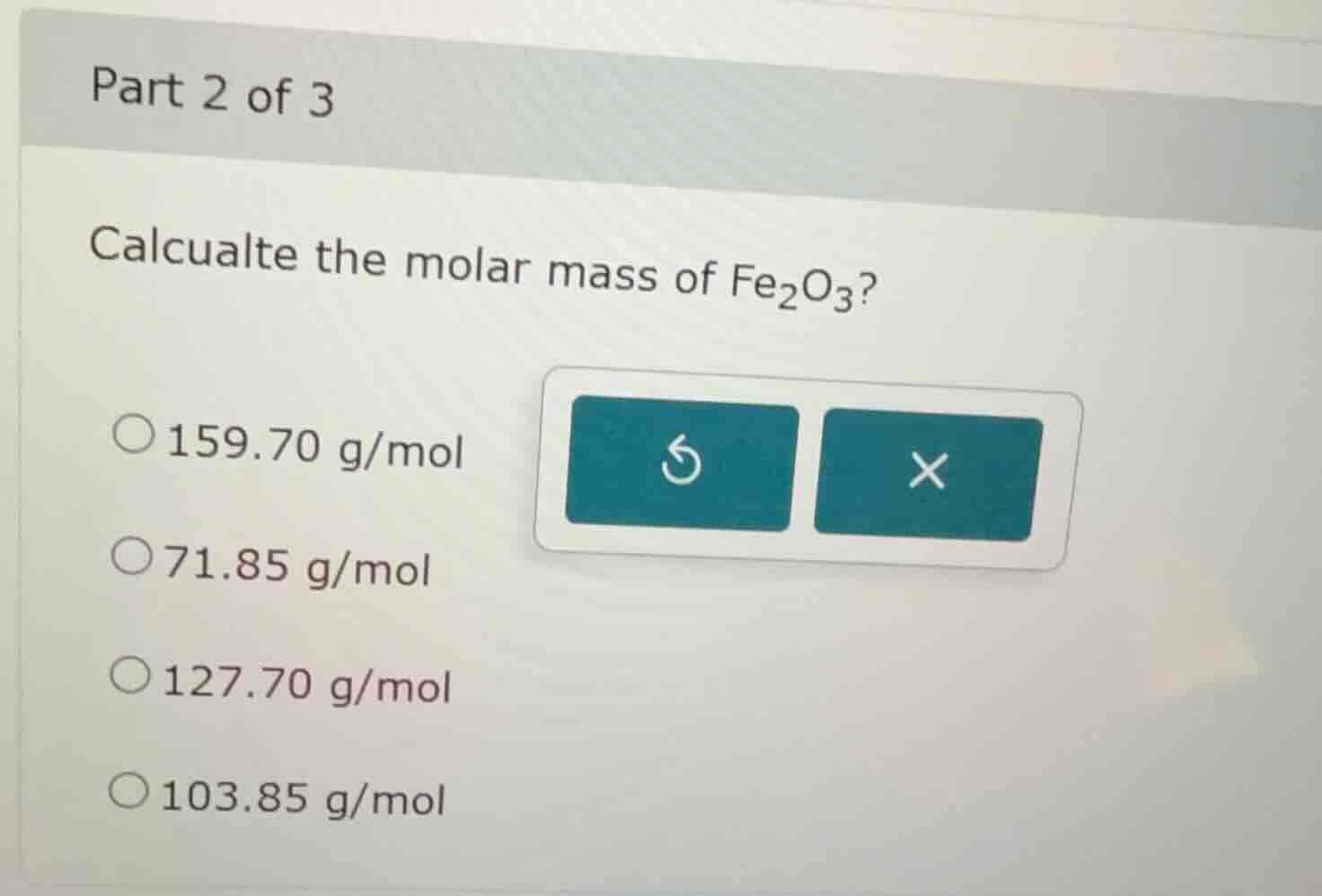
part 2 of 3
calcualte the molar mass of fe₂o₃?
○159.70 g/mol
○71.85 g/mol
○127.70 g/mol
○103.85 g/mol
Step1: Find atomic masses
Atomic mass of Fe: $55.85$ g/mol, O: $16.00$ g/mol.
Step2: Calculate Fe contribution
For $Fe_2$: $2\times55.85 = 111.70$ g/mol.
Step3: Calculate O contribution
For $O_3$: $3\times16.00 = 48.00$ g/mol.
Step4: Sum contributions
Molar mass = $111.70 + 48.00 = 159.70$ g/mol.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
159.70 g/mol (corresponding to the option: 159.70 g/mol)