QUESTION IMAGE
Question
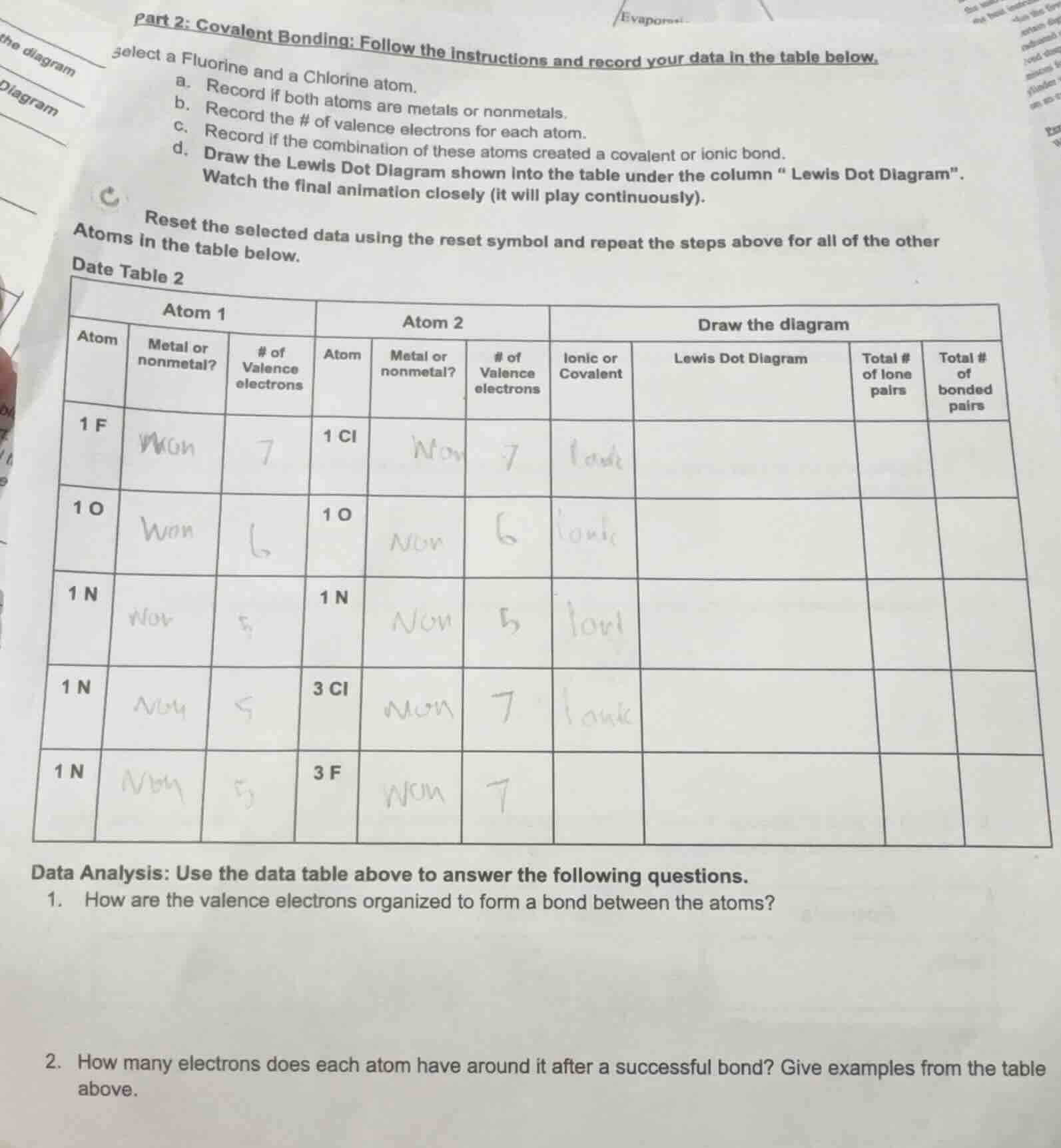
part 2: covalent bonding: follow the instructions and record your data in the table below. select a fluorine and a chlorine atom. a. record if both atoms are metals or nonmetals. b. record the # of valence electrons for each atom. c. record if the combination of these atoms created a covalent or ionic bond. d. draw the lewis dot diagram shown into the table under the column “lewis dot diagram”. watch the final animation closely (it will play continuously). reset the selected data using the reset symbol and repeat the steps above for all of the other atoms in the table below. data table 2 table with columns atom 1 (atom, metal or nonmetal?, # of valence electrons), atom 2 (atom, metal or nonmetal?, # of valence electrons), draw the diagram (ionic or covalent, lewis dot diagram, total # of lone pairs, total # of bonded pairs) and rows for 1 f & 1 cl, 1 o & 1 o, 1 n & 1 n, 1 n & 3 cl, 1 n & 3 f with some handwritten entries data analysis: use the data table above to answer the following questions. 1. how are the valence electrons organized to form a bond between the atoms? 2. how many electrons does each atom have around it after a successful bond? give examples from the table above.
- For covalent bonding (all pairs here are nonmetals), valence electrons are shared between atoms to fill each atom's outer valence shell (to reach a stable octet, or duet for hydrogen, not present here). Pairs of electrons are either shared (bonded pairs) or remain as unshared lone pairs on individual atoms.
- After bonding, each atom has 8 valence electrons (octet rule, stable configuration) around it. For example:
- F in F-Cl: 6 lone electrons (3 lone pairs) + 2 shared electrons (1 bonded pair) = 8 total electrons
- O in O=O: 4 lone electrons (2 lone pairs) + 4 shared electrons (2 bonded pairs) = 8 total electrons
- N in N≡N: 2 lone electrons (1 lone pair) + 6 shared electrons (3 bonded pairs) = 8 total electrons
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Valence electrons are shared: some form shared bonded pairs between atoms, while others stay as unshared lone pairs on each atom, to fill each atom's valence shell for stability.
- Each atom has 8 valence electrons around it (following the octet rule).
Examples:
- In the F-Cl pair: Each F and Cl has 8 electrons (6 as lone pairs, 2 shared in a single bond).
- In the O-O pair: Each O has 8 electrons (4 as lone pairs, 4 shared in a double bond).
- In the N-N pair: Each N has 8 electrons (2 as a lone pair, 6 shared in a triple bond).