QUESTION IMAGE
Question
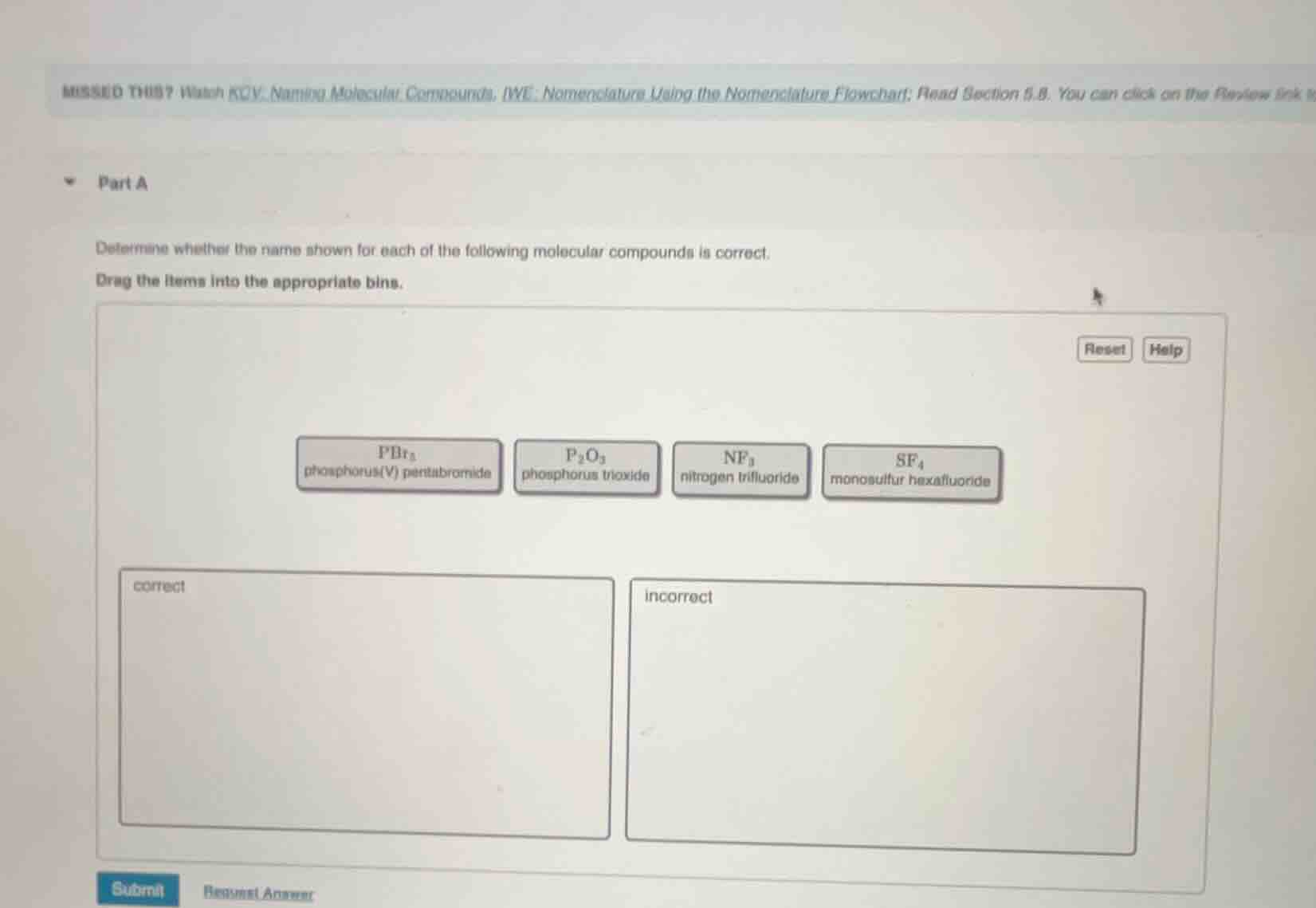
part a
determine whether the name shown for each of the following molecular compounds is correct.
drag the items into the appropriate bins.
pbr₅ phosphorus(v) pentabromide
p₂o₃ phosphorus trioxide
nf₃ nitrogen trifluoride
sf₄ monosulfur hexafluoride
To solve this, we analyze each compound's naming using molecular compound nomenclature (prefixes for non - metal - non - metal compounds, and for some with variable oxidation states, the Stock system for phosphorus in some cases):
1. Analyze \( \ce{PBr_5} \) (phosphorus(V) pentabromide)
- For \( \ce{PBr_5} \), phosphorus has a +5 oxidation state (since each Br is - 1, and \( 5\times(- 1)+x = 0\Rightarrow x = + 5\)). The Stock system (using Roman numerals) can be used here, and the prefix "penta - " for 5 bromine atoms. So the name "phosphorus(V) pentabromide" is correct.
2. Analyze \( \ce{P_2O_3} \) (phosphorus trioxide)
- In \( \ce{P_2O_3} \), we use prefixes. The subscript of P is 2 (so "di - ") and O is 3 (so "tri - "). The correct name should be "diphosphorus trioxide". The given name "phosphorus trioxide" is incorrect as it misses the prefix "di - " for phosphorus.
3. Analyze \( \ce{NF_3} \) (nitrogen trifluoride)
- For \( \ce{NF_3} \), N (nitrogen) has a subscript of 1 (no prefix needed, as "mono - " is only used for the second element in most cases) and F (fluorine) has a subscript of 3 (prefix "tri - "). So the name "nitrogen trifluoride" is correct.
4. Analyze \( \ce{SF_4} \) (monosulfur hexafluoride)
- In \( \ce{SF_4} \), S has a subscript of 1 (we don't use "mono - " for the first element in molecular compound naming) and F has a subscript of 4 (prefix "tetra - ", not "hexa - "). The correct name should be "sulfur tetrafluoride". So the name "monosulfur hexafluoride" is incorrect.
Correct Bins:
- Correct: \( \ce{PBr_5} \) (phosphorus(V) pentabromide), \( \ce{NF_3} \) (nitrogen trifluoride)
- Incorrect: \( \ce{P_2O_3} \) (phosphorus trioxide), \( \ce{SF_4} \) (monosulfur hexafluoride)
So when dragging the items:
- Put " \( \ce{PBr_5} \): phosphorus(V) pentabromide" and " \( \ce{NF_3} \): nitrogen trifluoride" into the "correct" bin.
- Put " \( \ce{P_2O_3} \): phosphorus trioxide" and " \( \ce{SF_4} \): monosulfur hexafluoride" into the "incorrect" bin.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
To solve this, we analyze each compound's naming using molecular compound nomenclature (prefixes for non - metal - non - metal compounds, and for some with variable oxidation states, the Stock system for phosphorus in some cases):
1. Analyze \( \ce{PBr_5} \) (phosphorus(V) pentabromide)
- For \( \ce{PBr_5} \), phosphorus has a +5 oxidation state (since each Br is - 1, and \( 5\times(- 1)+x = 0\Rightarrow x = + 5\)). The Stock system (using Roman numerals) can be used here, and the prefix "penta - " for 5 bromine atoms. So the name "phosphorus(V) pentabromide" is correct.
2. Analyze \( \ce{P_2O_3} \) (phosphorus trioxide)
- In \( \ce{P_2O_3} \), we use prefixes. The subscript of P is 2 (so "di - ") and O is 3 (so "tri - "). The correct name should be "diphosphorus trioxide". The given name "phosphorus trioxide" is incorrect as it misses the prefix "di - " for phosphorus.
3. Analyze \( \ce{NF_3} \) (nitrogen trifluoride)
- For \( \ce{NF_3} \), N (nitrogen) has a subscript of 1 (no prefix needed, as "mono - " is only used for the second element in most cases) and F (fluorine) has a subscript of 3 (prefix "tri - "). So the name "nitrogen trifluoride" is correct.
4. Analyze \( \ce{SF_4} \) (monosulfur hexafluoride)
- In \( \ce{SF_4} \), S has a subscript of 1 (we don't use "mono - " for the first element in molecular compound naming) and F has a subscript of 4 (prefix "tetra - ", not "hexa - "). The correct name should be "sulfur tetrafluoride". So the name "monosulfur hexafluoride" is incorrect.
Correct Bins:
- Correct: \( \ce{PBr_5} \) (phosphorus(V) pentabromide), \( \ce{NF_3} \) (nitrogen trifluoride)
- Incorrect: \( \ce{P_2O_3} \) (phosphorus trioxide), \( \ce{SF_4} \) (monosulfur hexafluoride)
So when dragging the items:
- Put " \( \ce{PBr_5} \): phosphorus(V) pentabromide" and " \( \ce{NF_3} \): nitrogen trifluoride" into the "correct" bin.
- Put " \( \ce{P_2O_3} \): phosphorus trioxide" and " \( \ce{SF_4} \): monosulfur hexafluoride" into the "incorrect" bin.