QUESTION IMAGE
Question
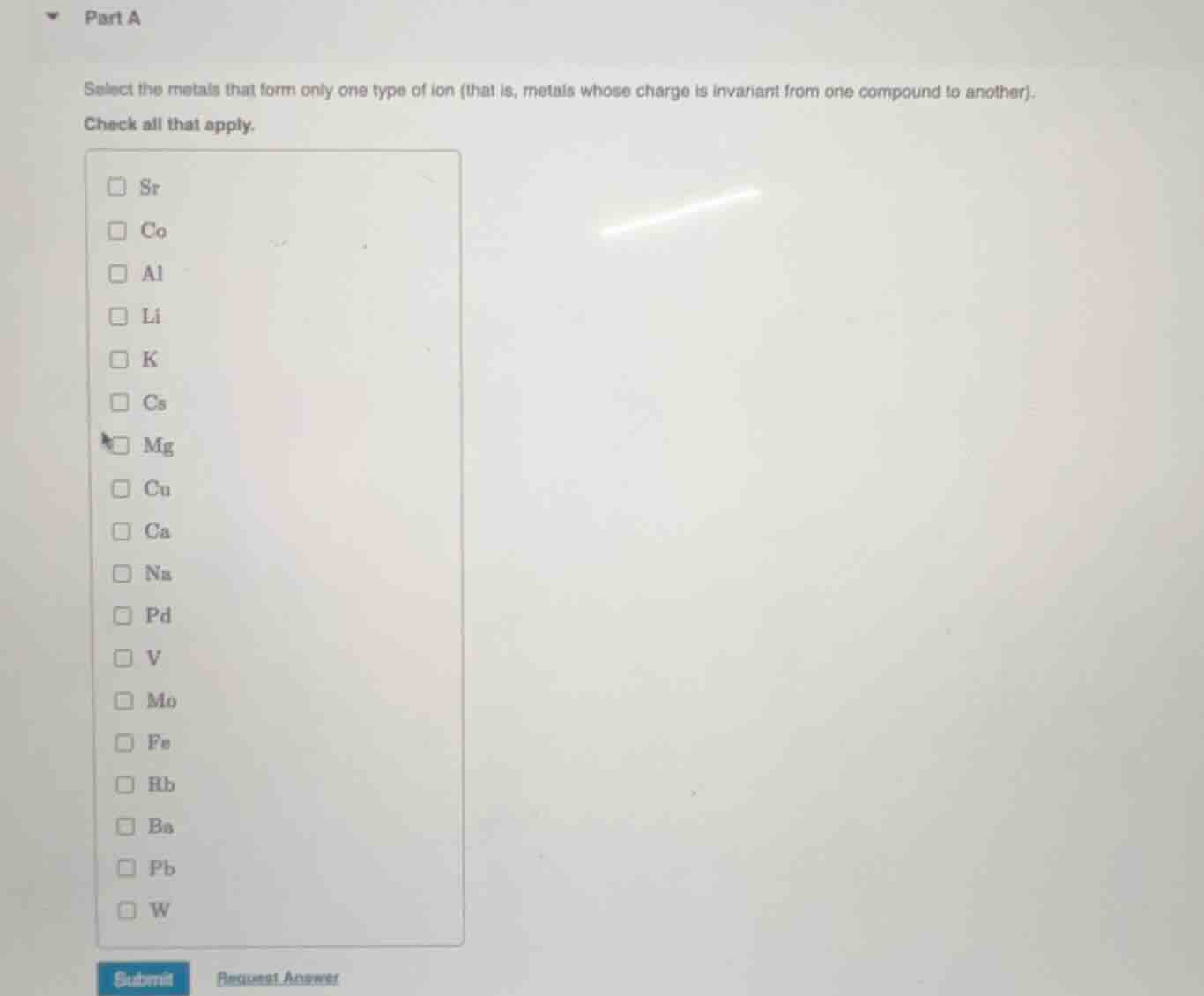
part a
select the metals that form only one type of ion (that is, metals whose charge is invariant from one compound to another).
check all that apply.
sr
co
al
li
k
cs
mg
cu
ca
na
pd
v
mo
fe
rb
ba
pb
w
To determine metals that form only one type of ion, we refer to the concept of fixed oxidation states. Alkali metals (Group 1: Li, K, Cs, Na, Rb) have a fixed +1 charge. Alkaline earth metals (Group 2: Sr, Mg, Ca, Ba) have a fixed +2 charge. Aluminum (Al) has a fixed +3 charge. Transition metals (Co, Cu, Pd, V, Mo, Fe, Pb, W) can have variable oxidation states. So the metals with invariant charge are Sr, Al, Li, K, Cs, Mg, Ca, Na, Rb, Ba.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. Sr, B. Al, C. Li, D. K, E. Cs, F. Mg, G. Ca, H. Na, I. Rb, J. Ba