QUESTION IMAGE
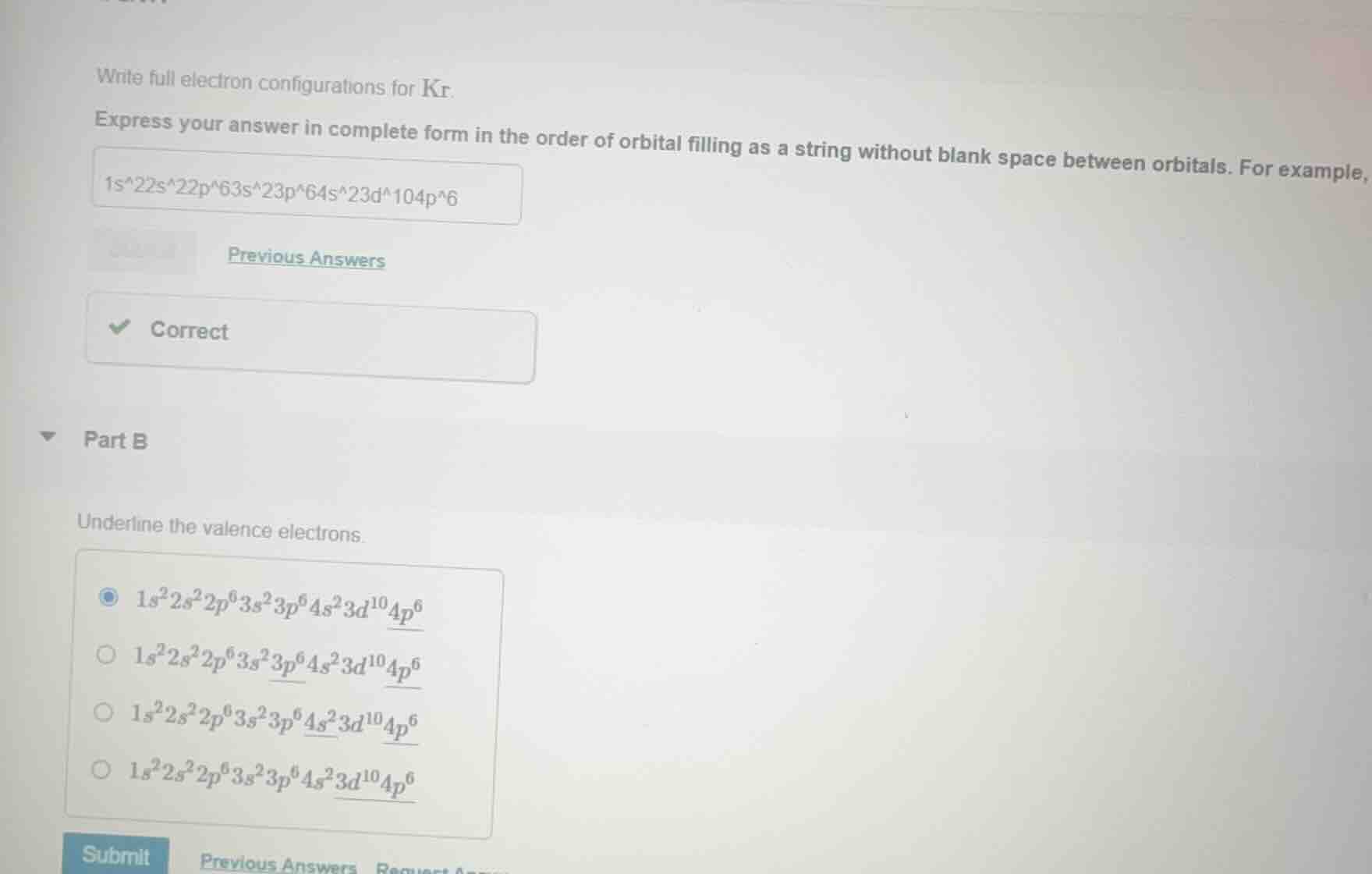
Question
part b
underline the valence electrons.
- $1s^2 2s^2 2p^6 3s^2 3p^6 4s^2 3d^{10} \underline{4p^6}$
- $1s^2 2s^2 2p^6 3s^2 \underline{3p^6} 4s^2 3d^{10} \underline{4p^6}$
- $1s^2 2s^2 2p^6 3s^2 3p^6 \underline{4s^2} 3d^{10} \underline{4p^6}$
- $1s^2 2s^2 2p^6 3s^2 3p^6 4s^2 \underline{3d^{10}} \underline{4p^6}$
Valence electrons are the electrons in the outermost principal energy level of an atom. For krypton (Kr), which is a noble gas in the 4th period, its outermost principal energy level is n=4, containing the $4s^2$ and $4p^6$ orbitals. However, in the given options, the correct underlining targets the outermost valence shell electrons, which are the $4p^6$ paired with the fact that for transition metals, the ns electrons are also valence, but Kr's full valence shell is $4s^24p^6$, and the correct option underlines the $4p^6$ as part of the valence electrons (the option that correctly identifies the outermost electrons as valence).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. $1s^22s^22p^63s^23p^64s^23d^{10}\underline{4p^6}$