QUESTION IMAGE
Question
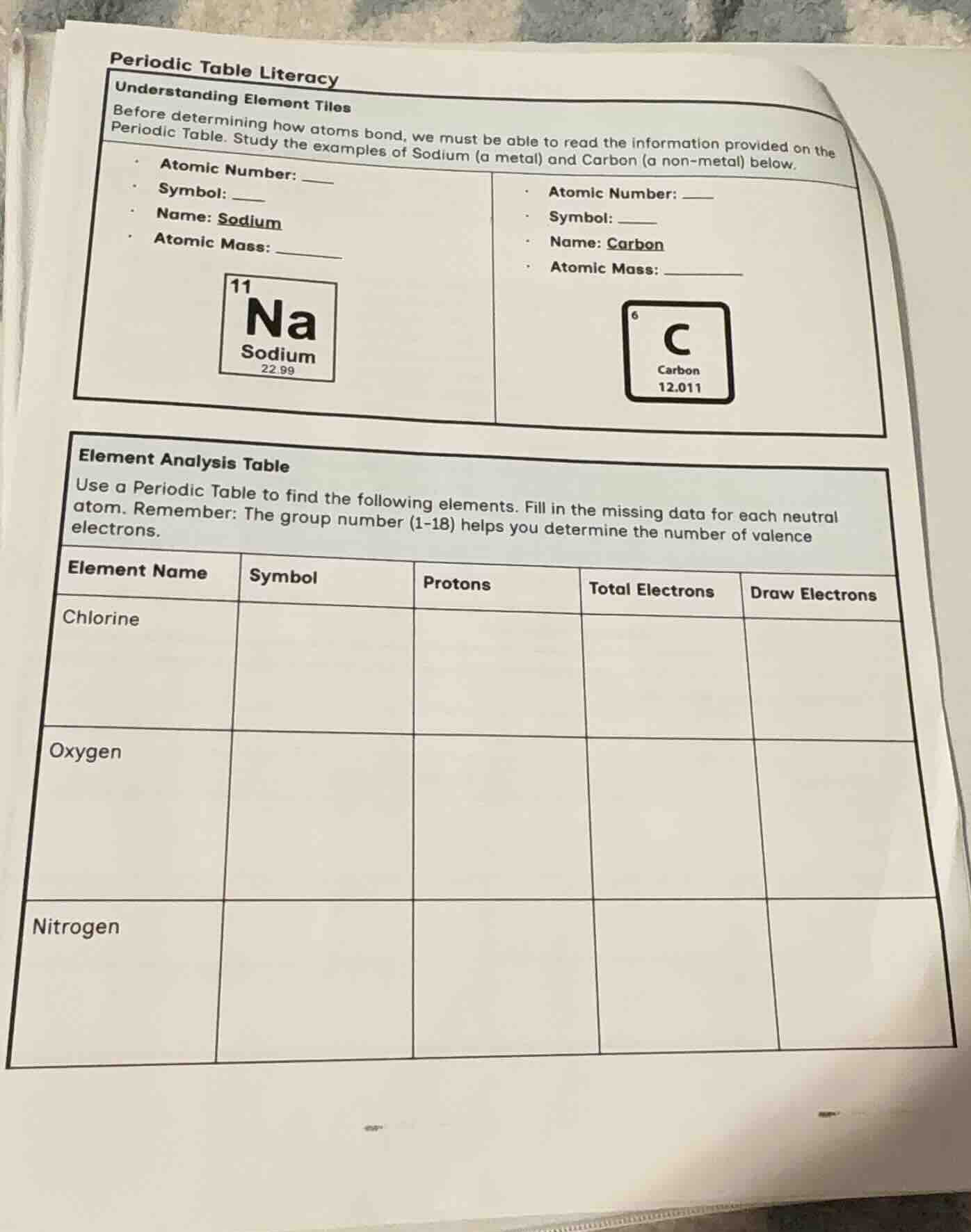
periodic table literacy
understanding element tiles
before determining how atoms bond, we must be able to read the information provided on the
periodic table. study the examples of sodium (a metal) and carbon (a non-metal) below.
· atomic number: ____
· symbol: ____
· name: sodium
· atomic mass: _______
· atomic number: ____
· symbol: ____
· name: carbon
· atomic mass: _______
element analysis table
use a periodic table to find the following elements. fill in the missing data for each neutral
atom. remember: the group number (1-18) helps you determine the number of valence
electrons.
element name | symbol | protons | total electrons | draw electrons
chlorine | | | |
oxygen | | | |
nitrogen | | | |
Step1: Fill Sodium tile data
Atomic Number: $11$
Symbol: $\text{Na}$
Atomic Mass: $22.99$
Step2: Fill Carbon tile data
Atomic Number: $6$
Symbol: $\text{C}$
Atomic Mass: $12.011$
Step3: Fill Chlorine table data
Symbol: $\text{Cl}$
Protons: $17$ (equal to atomic number)
Total Electrons: $17$ (neutral atom: electrons = protons)
Electron drawing: 2 electrons in first shell, 8 in second, 7 in third
Step4: Fill Oxygen table data
Symbol: $\text{O}$
Protons: $8$ (equal to atomic number)
Total Electrons: $8$ (neutral atom: electrons = protons)
Electron drawing: 2 electrons in first shell, 6 in second
Step5: Fill Nitrogen table data
Symbol: $\text{N}$
Protons: $7$ (equal to atomic number)
Total Electrons: $7$ (neutral atom: electrons = protons)
Electron drawing: 2 electrons in first shell, 5 in second
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Understanding Element Tiles
Sodium
- Atomic Number: $11$
- Symbol: $\text{Na}$
- Atomic Mass: $22.99$
Carbon
- Atomic Number: $6$
- Symbol: $\text{C}$
- Atomic Mass: $12.011$
Element Analysis Table
| Element Name | Symbol | Protons | Total Electrons | Draw Electrons |
|---|---|---|---|---|
| Oxygen | $\text{O}$ | $8$ | $8$ | 2 electrons (1st shell), 6 electrons (2nd shell) |
| Nitrogen | $\text{N}$ | $7$ | $7$ | 2 electrons (1st shell), 5 electrons (2nd shell) |