QUESTION IMAGE
Question
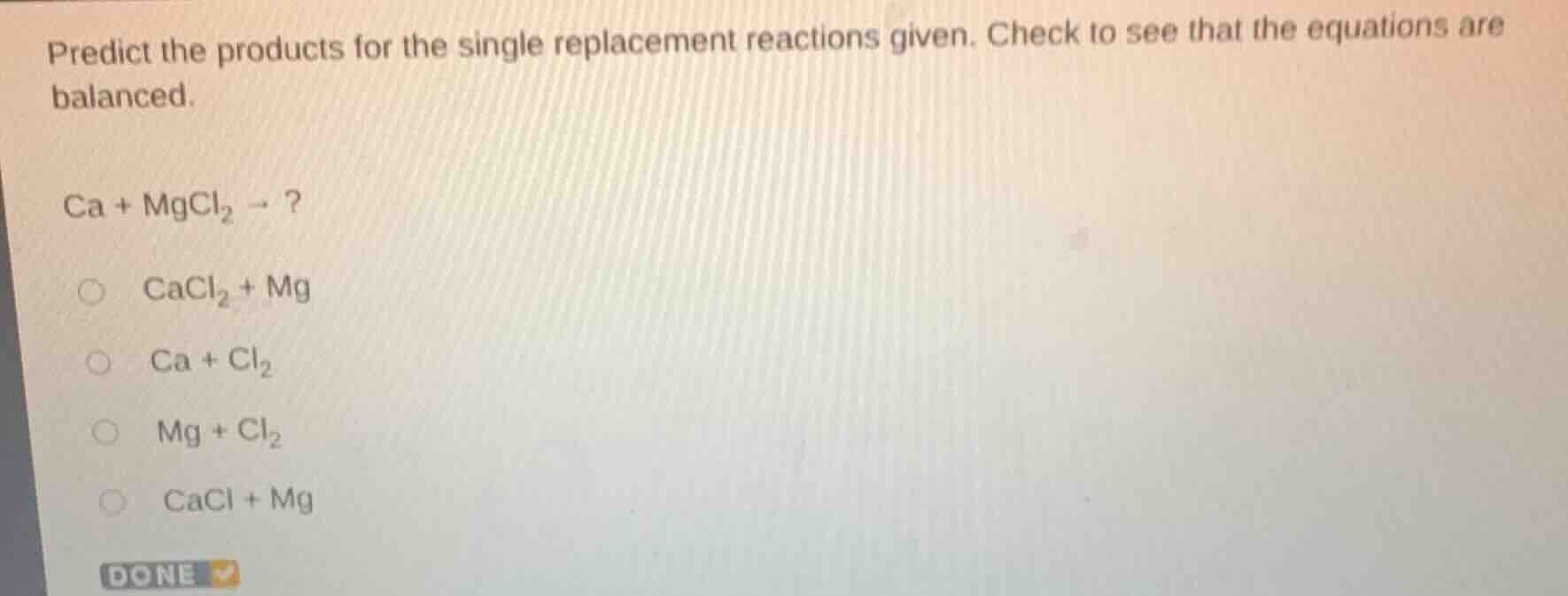
predict the products for the single replacement reactions given. check to see that the equations are balanced.
ca + mgcl₂ → ?
cacl₂ + mg
ca + cl₂
mg + cl₂
cacl + mg
In a single - replacement reaction, a more reactive metal can replace a less reactive metal in a compound. Calcium (Ca) is more reactive than magnesium (Mg) according to the activity series of metals. So, Ca will replace Mg in $MgCl_2$. The general form of a single - replacement reaction for metals is $A + BC
ightarrow AC + B$, where A is the more reactive metal, B is the less reactive metal, and C is the non - metal (in this case, Cl). So when Ca reacts with $MgCl_2$, Ca will combine with $Cl^-$ ions and Mg will be displaced. The formula of calcium chloride is $CaCl_2$ (since Ca has a + 2 charge and Cl has a - 1 charge, so 1 Ca²⁺ combines with 2 Cl⁻ to form $CaCl_2$) and the displaced metal is Mg. So the reaction is $Ca+MgCl_2
ightarrow CaCl_2 + Mg$. We also check the balance of the equation: on the left side, we have 1 Ca, 1 Mg, and 2 Cl. On the right side, we have 1 Ca, 1 Mg, and 2 Cl (in $CaCl_2$), so the equation is balanced.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. $CaCl_2 + Mg$