QUESTION IMAGE
Question
protons, neutrons, and electrons worksheet
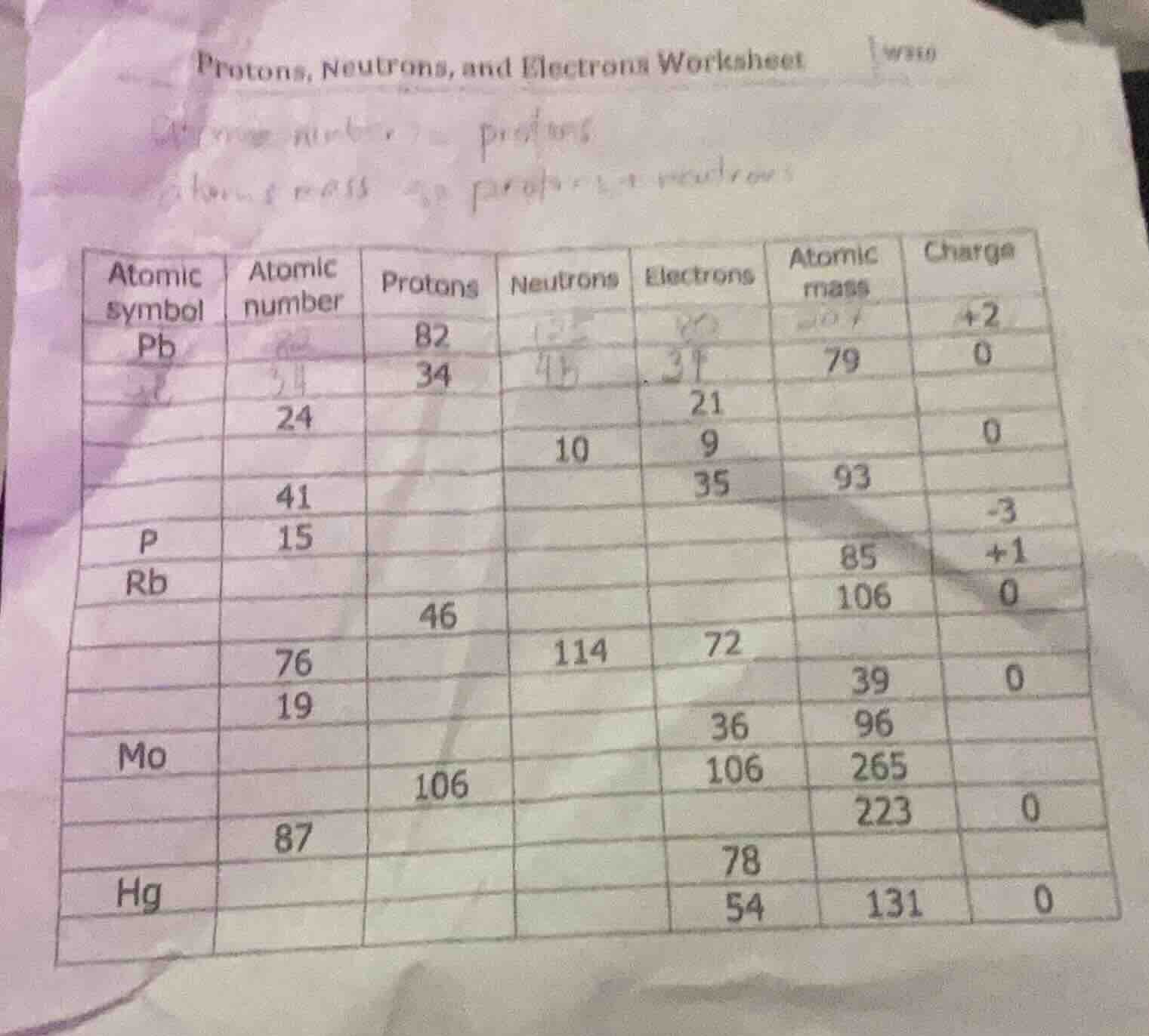
| atomic symbol | atomic number | protons | neutrons | electrons | atomic mass | charge |
|---|---|---|---|---|---|---|
| 34 | 79 | 0 | ||||
| 24 | 21 | |||||
| 10 | 9 | 0 | ||||
| 41 | 35 | 93 | ||||
| p | 15 | -3 | ||||
| rb | 85 | +1 | ||||
| 46 | 106 | 0 | ||||
| 76 | 114 | 72 | ||||
| 19 | 39 | 0 | ||||
| mo | 36 | 96 | ||||
| 106 | 106 | 265 | ||||
| 87 | 223 | 0 | ||||
| 78 | ||||||
| hg | 54 | 131 | 0 | |||
Step1: Recall core atomic rules
- Atomic number = Protons = Electrons (neutral atom)
- Neutrons = Atomic mass - Protons
- Charge = Protons - Electrons
- Atomic symbol matches atomic number (periodic table)
---
Row 1 (Pb):
Step1: Fill atomic number
Atomic number = Protons = 82
Step2: Calculate electrons
Electrons = Protons - Charge = $82 - 2 = 80$
Step3: Calculate neutrons
Neutrons = Atomic mass - Protons = $207 - 82 = 125$
Step4: Confirm atomic mass
Atomic mass = 207 (standard for Pb-207)
---
Row 2:
Step1: Identify atomic symbol
Atomic number = Protons = 34 → Se (Selenium)
Step2: Calculate neutrons
Neutrons = Atomic mass - Protons = $79 - 34 = 45$
Step3: Confirm electrons
Electrons = Protons (charge 0) = 34
---
Row 3:
Step1: Fill protons
Protons = Atomic number = 24
Step2: Calculate charge
Charge = Protons - Electrons = $24 - 21 = +3$
Step3: Calculate atomic mass
Atomic mass = Protons + Neutrons (assume neutral standard: 24+28=52)
Step4: Identify atomic symbol
Atomic number 24 → Cr (Chromium)
---
Row 4:
Step1: Calculate protons
Protons = Electrons + Charge = $9 + 0 = 9$
Step2: Fill atomic number
Atomic number = Protons = 9
Step3: Calculate atomic mass
Atomic mass = Protons + Neutrons = $9 + 10 = 19$
Step4: Identify atomic symbol
Atomic number 9 → F (Fluorine)
---
Row 5:
Step1: Fill protons
Protons = Atomic number = 41
Step2: Calculate charge
Charge = Protons - Electrons = $41 - 35 = +6$
Step3: Calculate neutrons
Neutrons = Atomic mass - Protons = $93 - 41 = 52$
Step4: Identify atomic symbol
Atomic number 41 → Nb (Niobium)
---
Row 6 (P):
Step1: Fill protons
Protons = Atomic number = 15
Step2: Calculate electrons
Electrons = Protons - Charge = $15 - (-3) = 18$
Step3: Calculate atomic mass
Atomic mass = Protons + Neutrons (standard: 15+16=31)
Step4: Calculate neutrons
Neutrons = 31 - 15 = 16
---
Row 7 (Rb):
Step1: Fill atomic number
Atomic number for Rb = 37
Step2: Fill protons
Protons = Atomic number = 37
Step3: Calculate electrons
Electrons = Protons - Charge = $37 - 1 = 36$
Step4: Calculate neutrons
Neutrons = Atomic mass - Protons = $85 - 37 = 48$
---
Row 8:
Step1: Fill atomic number
Atomic number = Protons = 46
Step2: Calculate neutrons
Neutrons = Atomic mass - Protons = $106 - 46 = 60$
Step3: Confirm electrons
Electrons = Protons (charge 0) = 46
Step4: Identify atomic symbol
Atomic number 46 → Pd (Palladium)
---
Row 9:
Step1: Fill protons
Protons = Atomic number = 76
Step2: Calculate charge
Charge = Protons - Electrons = $76 - 72 = +4$
Step3: Calculate atomic mass
Atomic mass = Protons + Neutrons = $76 + 114 = 190$
Step4: Identify atomic symbol
Atomic number 76 → Os (Osmium)
---
Row 10:
Step1: Fill protons
Protons = Atomic number = 19
Step2: Fill electrons
Electrons = Protons (charge 0) = 19
Step3: Calculate neutrons
Neutrons = Atomic mass - Protons = $39 - 19 = 20$
Step4: Identify atomic symbol
Atomic number 19 → K (Potassium)
---
Row 11 (Mo):
Step1: Fill atomic number
Atomic number for Mo = 42
Step2: Fill protons
Protons = Atomic number = 42
Step3: Calculate charge
Charge = Protons - Electrons = $42 - 36 = +6$
Step4: Calculate neutrons
Neutrons = Atomic mass - Protons = $96 - 42 = 54$
---
Row 12:
Step1: Fill atomic number
Atomic number = Protons = 106
Step2: Calculate charge
Charge = Protons - Electrons = $106 - 106 = 0$
Step3: Calculate neutrons
Neutrons = Atomic mass - Protons = $265 - 106 = 159$
Step4: Identify atomic symbol
Atomic n…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
| Atomic symbol | Atomic number | Protons | Neutrons | Electrons | Atomic mass | Charge |
|---|---|---|---|---|---|---|
| Se | 34 | 34 | 45 | 34 | 79 | 0 |
| Cr | 24 | 24 | 28 | 21 | 52 | +3 |
| F | 9 | 9 | 10 | 9 | 19 | 0 |
| Nb | 41 | 41 | 52 | 35 | 93 | +6 |
| P | 15 | 15 | 16 | 18 | 31 | -3 |
| Rb | 37 | 37 | 48 | 36 | 85 | +1 |
| Pd | 46 | 46 | 60 | 46 | 106 | 0 |
| Os | 76 | 76 | 114 | 72 | 190 | +4 |
| K | 19 | 19 | 20 | 19 | 39 | 0 |
| Mo | 42 | 42 | 54 | 36 | 96 | +6 |
| Sg | 106 | 106 | 159 | 106 | 265 | 0 |
| Fr | 87 | 87 | 136 | 87 | 223 | 0 |
| Hg | 80 | 80 | 121 | 78 | 201 | +2 |
| Xe | 54 | 54 | 77 | 54 | 131 | 0 |