QUESTION IMAGE
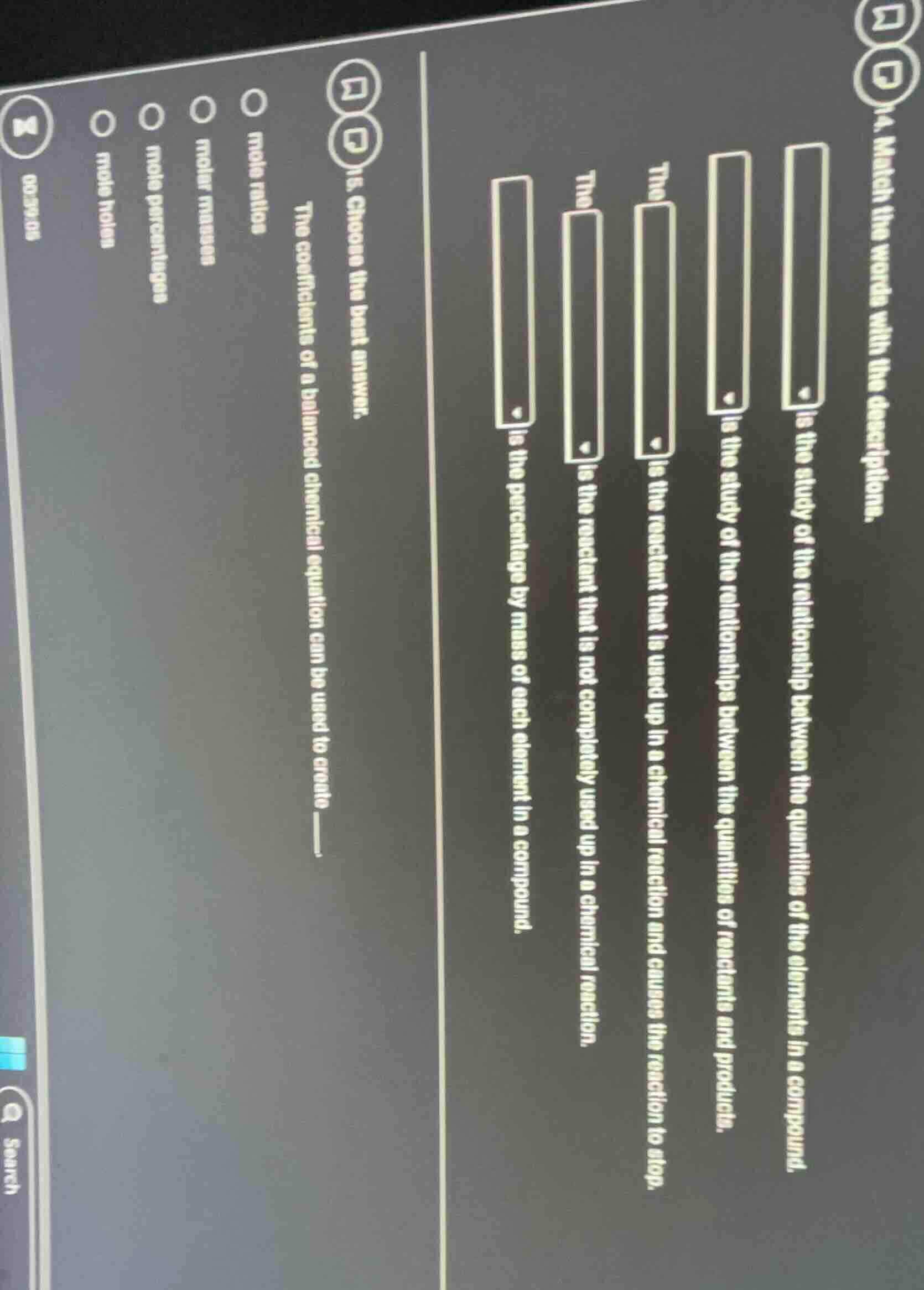
Question
q15. choose the best answer. the coefficients of a balanced chemical equation can be used to create ______.
options:
mole ratios
molar masses
mole percentages
mole holes
Brief Explanations
For Question 14:
- The study of element quantities in compounds is stoichiometry focused on composition, called composition stoichiometry.
- The study of reactant and product quantities in reactions is reaction stoichiometry.
- The reactant that runs out and stops the reaction is the limiting reactant.
- The reactant left unused after a reaction is the excess reactant.
- The mass percentage of an element in a compound is percent composition.
For Question 15:
Balanced chemical equation coefficients represent the relative number of moles of each substance, which defines mole ratios between reactants and products.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Question 14:
- composition stoichiometry is the study of the relationship between the quantities of the elements in a compound.
- reaction stoichiometry is the study of the relationships between the quantities of reactants and products.
- limiting reactant is the reactant that is used up in a chemical reaction and causes the reaction to stop.
- excess reactant is the reactant that is not completely used up in a chemical reaction.
- percent composition is the percentage by mass of each element in a compound.
Question 15:
mole ratios