QUESTION IMAGE
Question
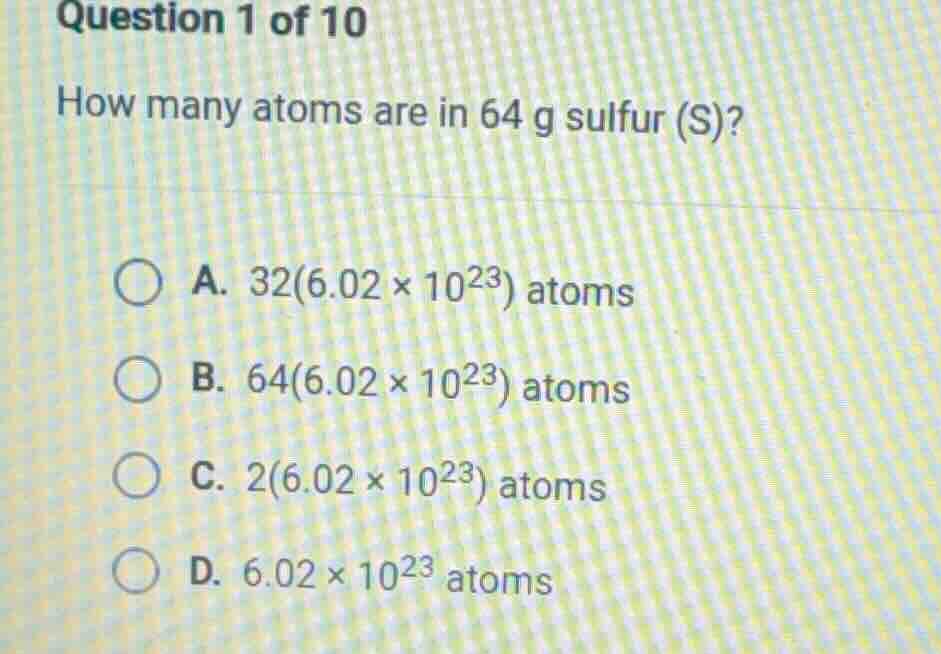
question 1 of 10
how many atoms are in 64 g sulfur (s)?
a. 32(6.02×10²³) atoms
b. 64(6.02×10²³) atoms
c. 2(6.02×10²³) atoms
d. 6.02×10²³ atoms
Step1: Find molar mass of S
Molar mass of S is 32 g/mol.
Step2: Calculate moles of S
Moles = mass / molar mass = 64 g / 32 g/mol = 2 mol.
Step3: Relate moles to atoms
1 mol has \(6.02\times10^{23}\) atoms, so 2 mol has \(2\times(6.02\times10^{23})\) atoms.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
C. \(2(6.02\times 10^{23})\) atoms