QUESTION IMAGE
Question
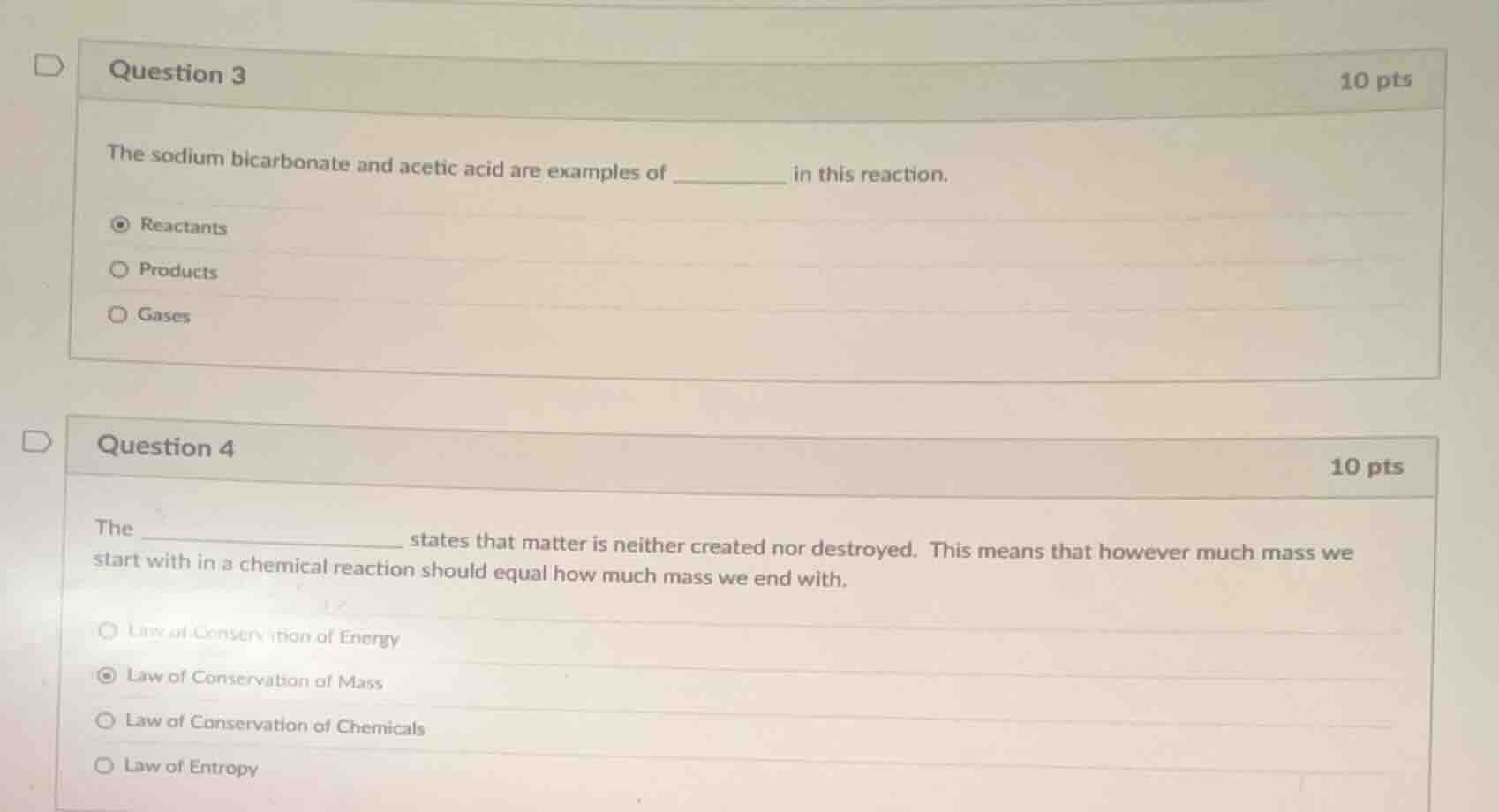
question 3
10 pts
the sodium bicarbonate and acetic acid are examples of _______ in this reaction.
- reactants
- products
- gases
question 4
10 pts
the ______________ states that matter is neither created nor destroyed. this means that however much mass we start with in a chemical reaction should equal how much mass we end with.
- law of conservation of energy
- law of conservation of mass
- law of conservation of chemicals
- law of entropy
For Question 3: In chemical reactions, substances that undergo a reaction to form new substances are called reactants. Sodium bicarbonate and acetic acid react with each other, so they are reactants.
For Question 4: The principle that matter is not created or destroyed in a chemical reaction, with total mass remaining constant, is defined by the Law of Conservation of Mass.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Question 3: A. Reactants
Question 4: B. Law of Conservation of Mass