QUESTION IMAGE
Question
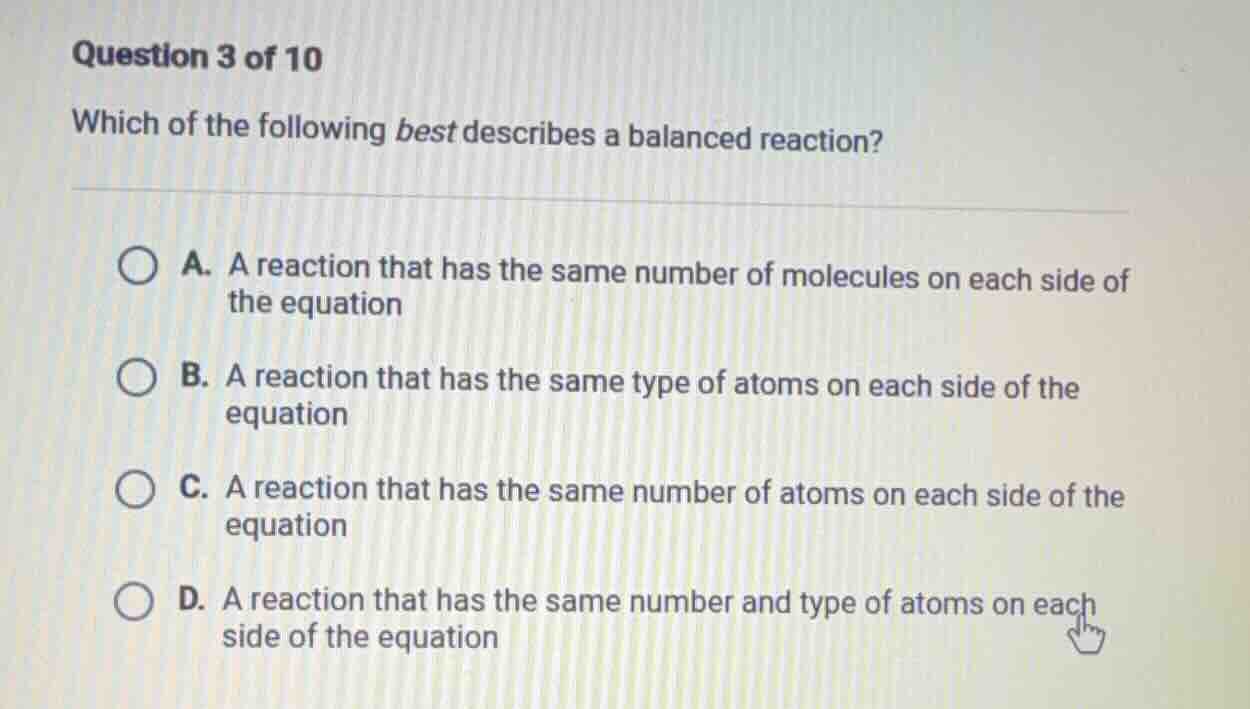
question 3 of 10
which of the following best describes a balanced reaction?
a. a reaction that has the same number of molecules on each side of
the equation
b. a reaction that has the same type of atoms on each side of the
equation
c. a reaction that has the same number of atoms on each side of the
equation
d. a reaction that has the same number and type of atoms on each
side of the equation
To determine the best description of a balanced chemical reaction, we analyze each option:
- Option A: Balancing a reaction is not about the number of molecules (molecules can be different in number but atoms are conserved). So A is incorrect.
- Option B: Just having the same type of atoms is not enough; the number of each type of atom must also be the same. So B is incorrect.
- Option C: Having the same number of atoms is insufficient if the types of atoms differ. So C is incorrect.
- Option D: A balanced chemical reaction follows the law of conservation of mass, which means the same number and type of atoms are present on both sides of the equation (reactants and products). This is the correct definition.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
D. A reaction that has the same number and type of atoms on each side of the equation