QUESTION IMAGE
Question
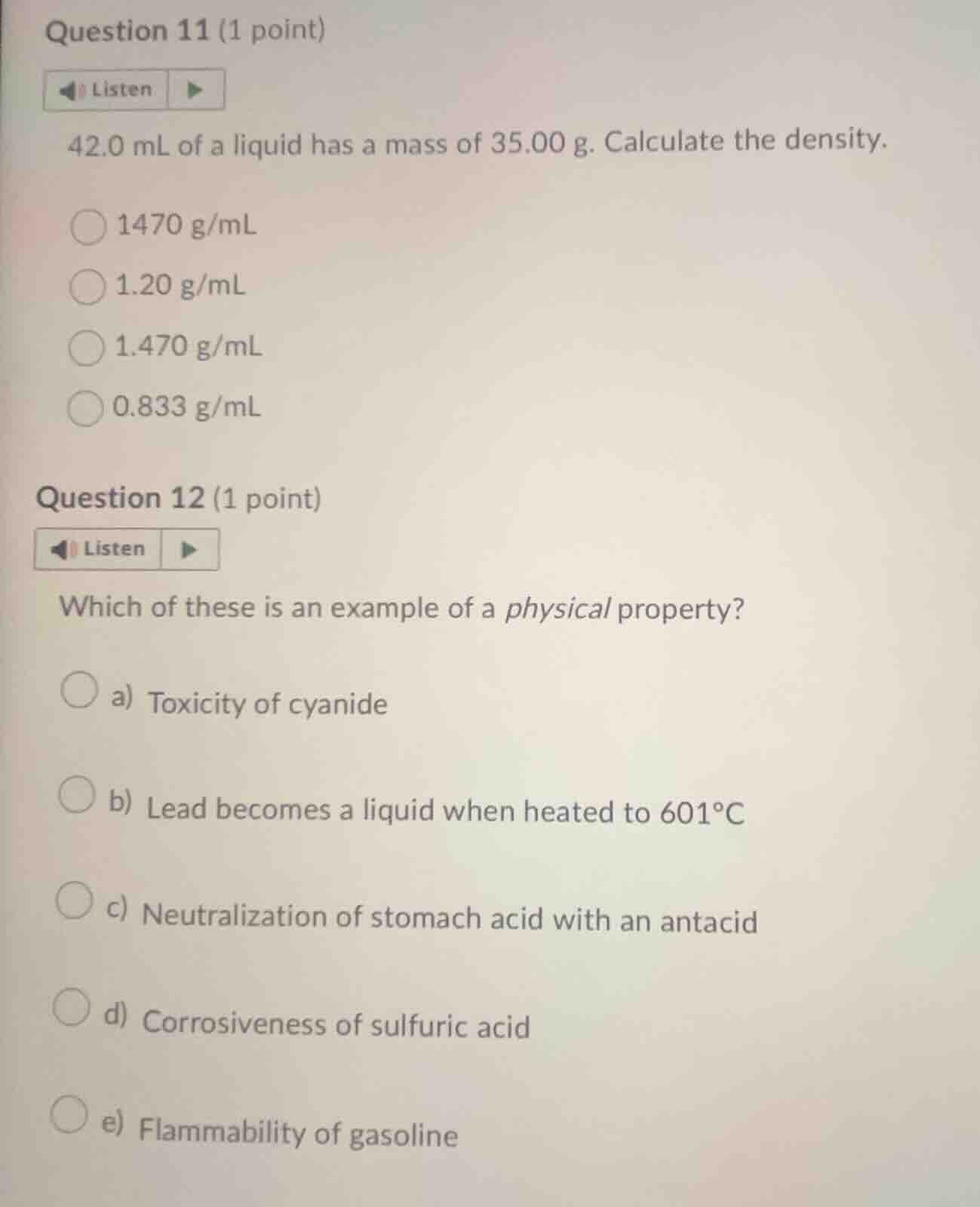
question 11 (1 point)
listen
42.0 ml of a liquid has a mass of 35.00 g. calculate the density.
1470 g/ml
1.20 g/ml
1.470 g/ml
0.833 g/ml
question 12 (1 point)
listen
which of these is an example of a physical property?
a) toxicity of cyanide
b) lead becomes a liquid when heated to 601°c
c) neutralization of stomach acid with an antacid
d) corrosiveness of sulfuric acid
e) flammability of gasoline
Question 11
Step1: Recall density formula
Density $
ho = \frac{\text{mass}}{\text{volume}}$
Step2: Substitute given values
$
ho = \frac{35.00\ \text{g}}{42.0\ \text{mL}}$
Step3: Calculate the result
$
ho = 0.833\ \text{g/mL}$
A physical property is a characteristic of a substance that can be observed or measured without changing the substance's chemical identity.
- Toxicity, neutralization, corrosiveness, and flammability all involve chemical changes or reactions.
- Melting of lead only changes its physical state (solid to liquid) without altering its chemical composition.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
0.833 g/mL
---