QUESTION IMAGE
Question
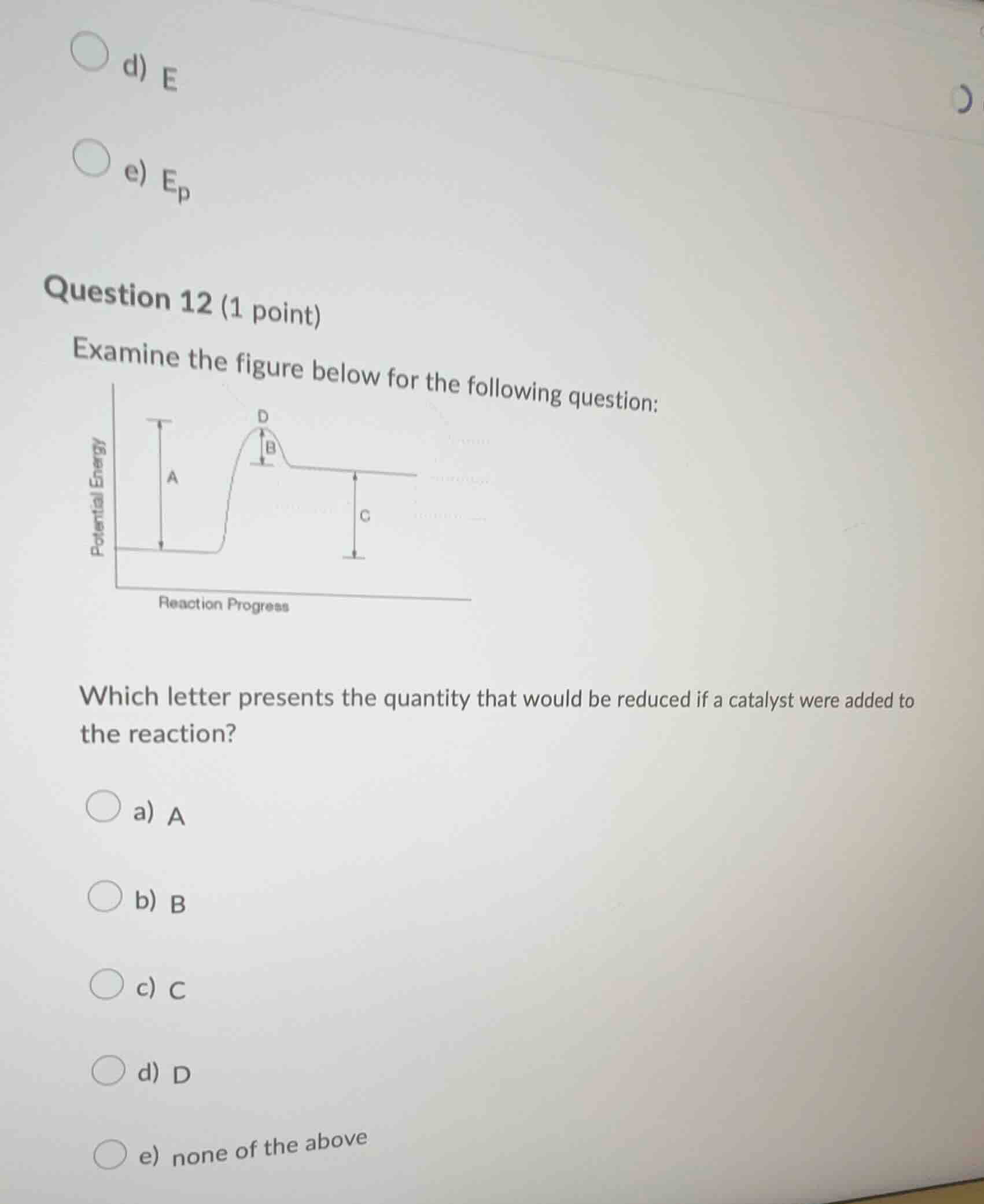
question 12 (1 point)
examine the figure below for the following question:
which letter presents the quantity that would be reduced if a catalyst were added to the reaction?
a) a
b) b
c) c
d) d
e) none of the above
A catalyst lowers the activation energy of a reaction. In the reaction - progress energy diagram, the activation energy is represented by the height of the energy barrier (the difference between the energy of the transition state and the reactants or intermediates). Here, the letter \( B \) represents the activation energy - related barrier that a catalyst would reduce. \( A \) and \( C \) represent the energy difference of reactants/products and other non - activation - energy related quantities, and \( D \) is the transition state energy point, not the activation energy value itself.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
b) B