QUESTION IMAGE
Question
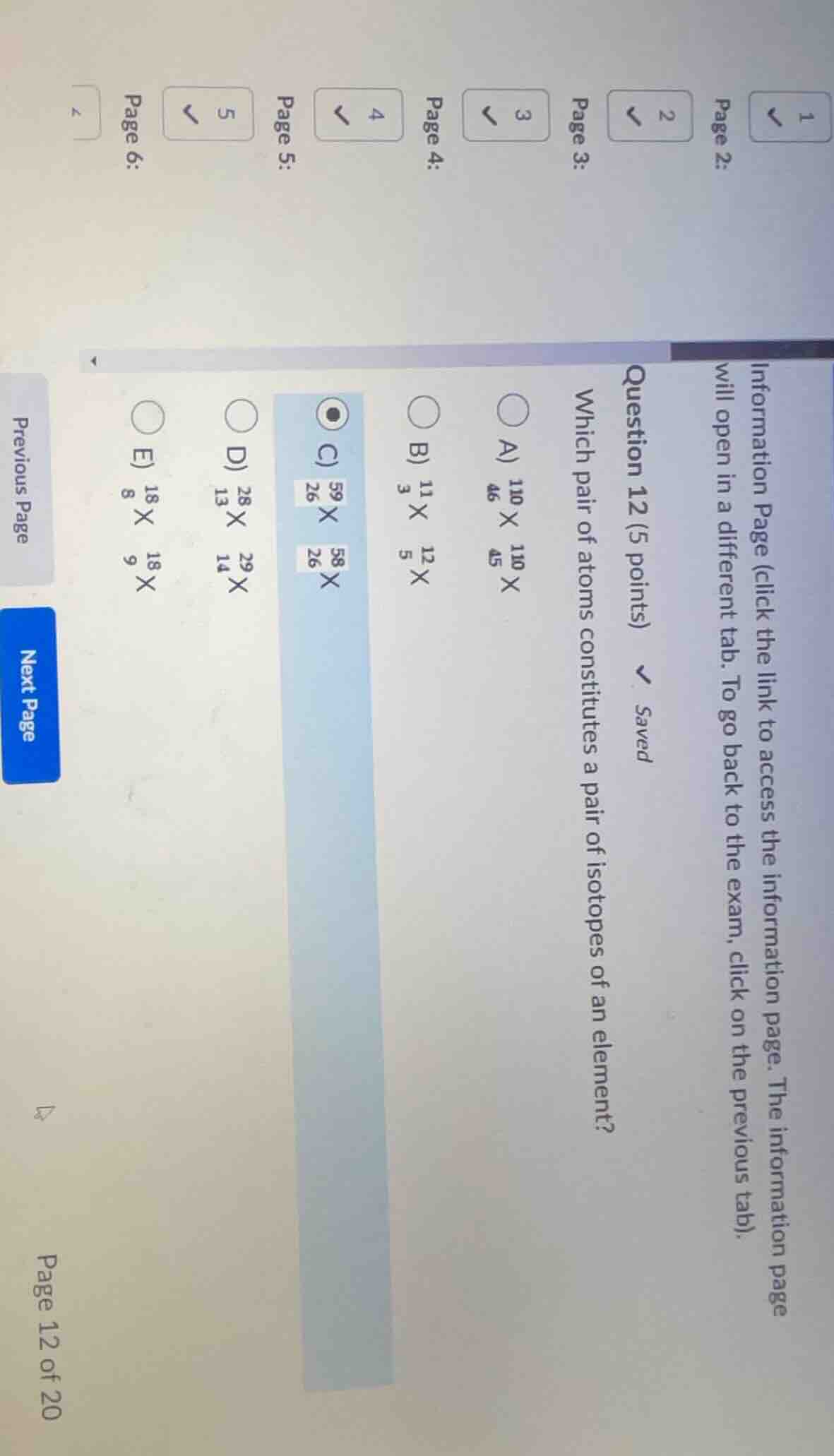
question 12 (5 points) \saved which pair of atoms constitutes a pair of isotopes of an element? a) \\(\ce{_{46}^{110}x}\\) \\(\ce{_{45}^{110}x}\\) b) \\(\ce{_{3}^{11}x}\\) \\(\ce{_{5}^{12}x}\\) c) \\(\ce{_{26}^{59}x}\\) \\(\ce{_{26}^{58}x}\\) d) \\(\ce{_{13}^{28}x}\\) \\(\ce{_{14}^{29}x}\\) e) \\(\ce{_{8}^{18}x}\\) \\(\ce{_{9}^{18}x}\\)
Isotopes are atoms of the same element, meaning they have the same atomic number (the bottom number, which represents the number of protons) but different mass numbers (the top number, which is protons + neutrons).
- Option A: Atomic numbers are 46 and 45 – different elements.
- Option B: Atomic numbers are 11 and 12 – different elements.
- Option C: Atomic number is 26 for both (same element, iron), mass numbers are 59 and 58 (different neutrons) – these are isotopes.
- Option D: Atomic numbers are 13 and 14 – different elements.
- Option E: Atomic numbers are 8 and 9 – different elements.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
C) $\boldsymbol{_{26}^{59}X}$ $\boldsymbol{_{26}^{58}X}$