QUESTION IMAGE
Question
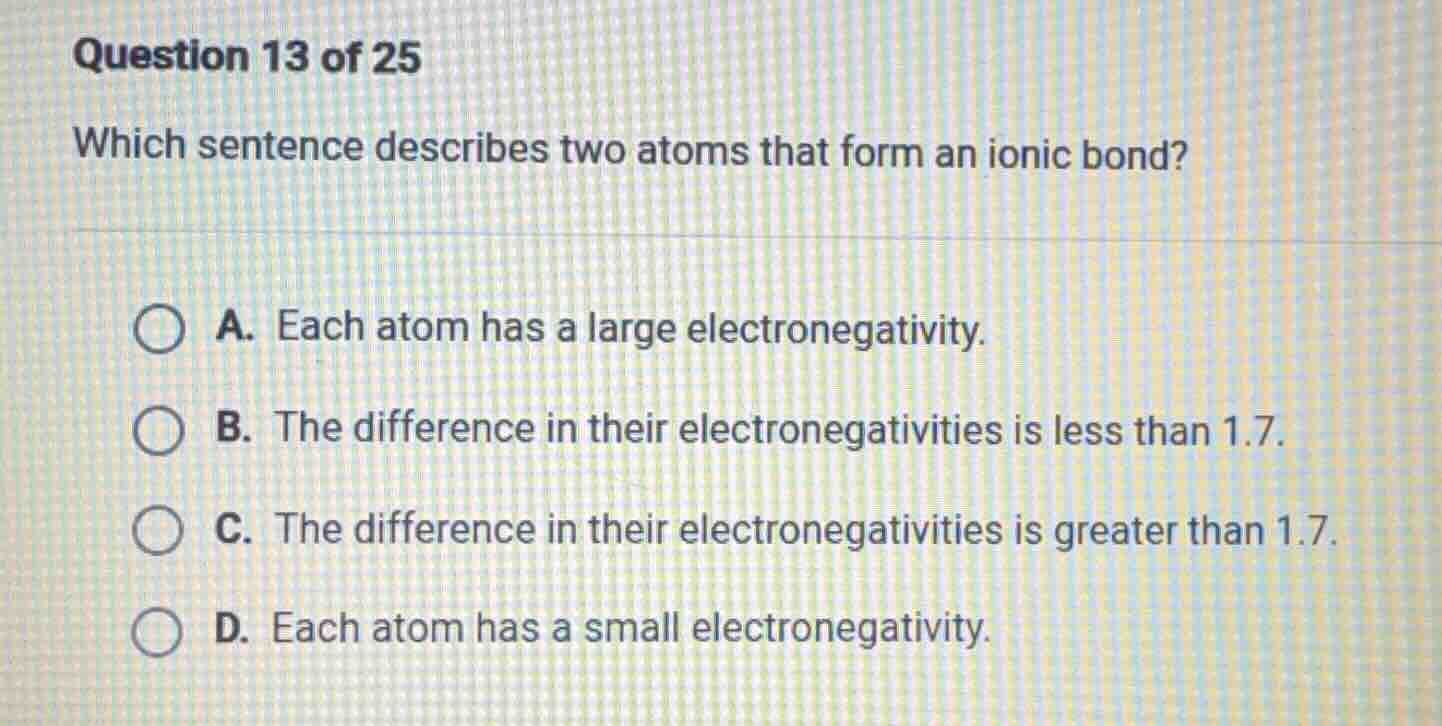
question 13 of 25
which sentence describes two atoms that form an ionic bond?
a. each atom has a large electronegativity.
b. the difference in their electronegativities is less than 1.7.
c. the difference in their electronegativities is greater than 1.7.
d. each atom has a small electronegativity.
To determine which sentence describes two atoms forming an ionic bond, we recall the concept of electronegativity and bond types. Ionic bonds typically form when the difference in electronegativity between two atoms is relatively large. A common guideline is that if the electronegativity difference is greater than 1.7, the bond is considered ionic.
- Option A: Having a large electronegativity for each atom doesn't necessarily mean they form an ionic bond (e.g., two highly electronegative atoms would likely form a covalent bond, like in \( \text{F}_2 \)).
- Option B: A difference less than 1.7 is more characteristic of a polar covalent bond, not an ionic bond.
- Option C: A difference greater than 1.7 aligns with the typical electronegativity difference for ionic bond formation.
- Option D: Having a small electronegativity for each atom doesn't indicate ionic bond formation (e.g., two atoms with low electronegativity would likely form a non - polar covalent bond, like in \( \text{H}_2 \)).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
C. The difference in their electronegativities is greater than 1.7.