QUESTION IMAGE
Question
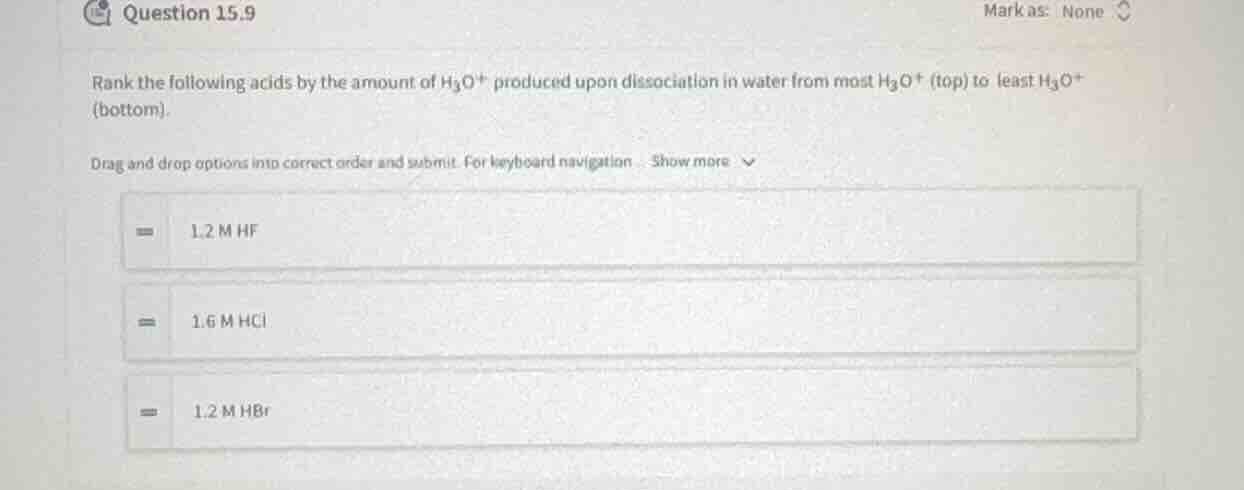
question 15.9
mark as: none
rank the following acids by the amount of $h_3o^+$ produced upon dissociation in water from most $h_3o^+$ (top) to least $h_3o^+$ (bottom).
drag and drop options into correct order and submit. for keyboard navigation show more
1.2 m hf
1.6 m hcl
1.2 m hbr
Step1: Classify acid strength
HCl and HBr are strong acids (complete dissociation); HF is a weak acid (partial dissociation, produces far less $\text{H}_3\text{O}^+$ at same concentration).
Step2: Compare strong acid concentrations
For strong acids, $\text{[H}_3\text{O}^+]$ equals acid concentration. 1.6 M HCl has higher concentration than 1.2 M HBr, so it produces more $\text{H}_3\text{O}^+$.
Step3: Rank all acids
Strong acids produce more $\text{H}_3\text{O}^+$ than weak acid; higher concentration strong acid > lower concentration strong acid > weak acid.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- 1.6 M HCl
- 1.2 M HBr
- 1.2 M HF