QUESTION IMAGE
Question
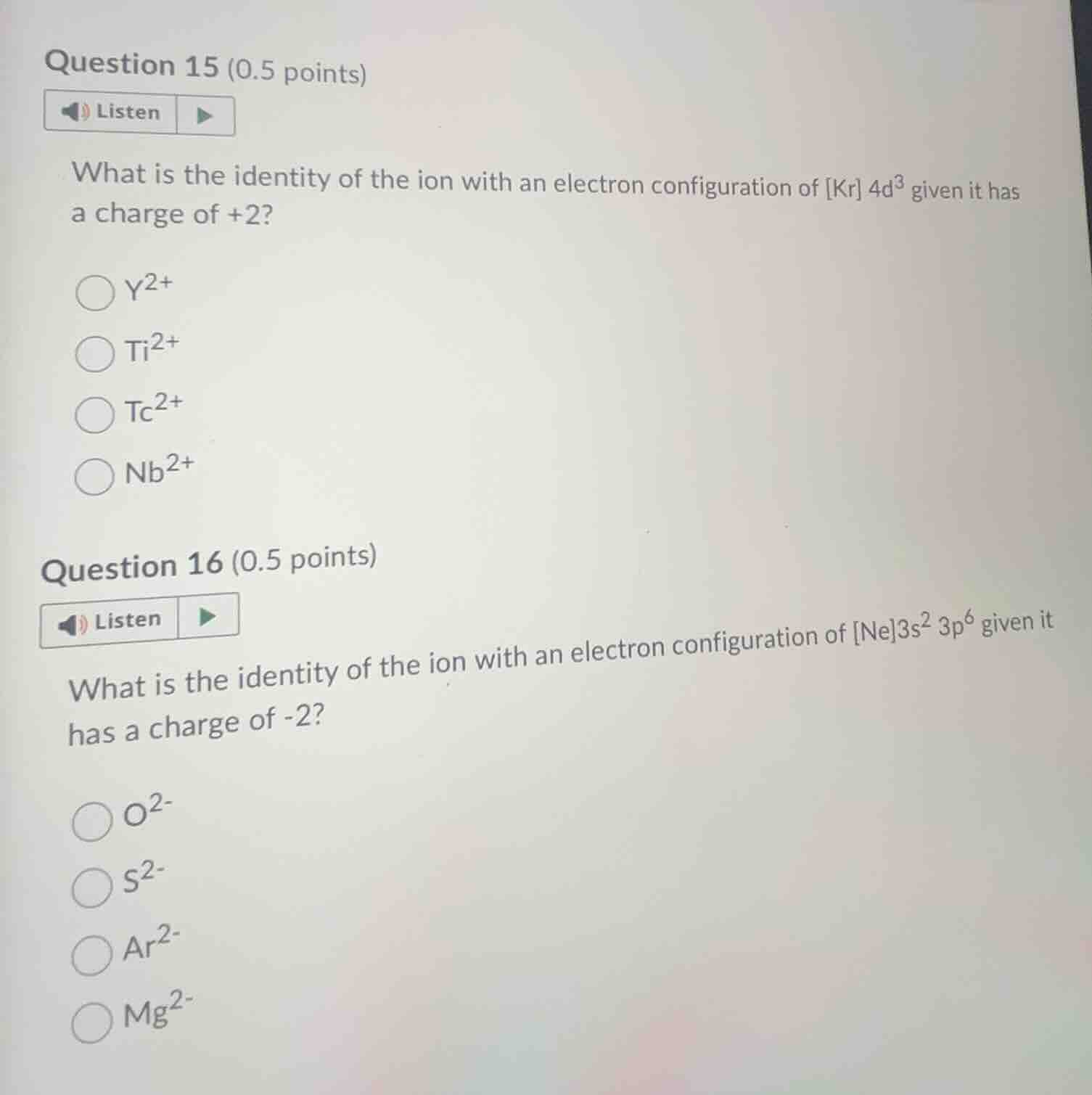
question 15 (0.5 points)
listen
what is the identity of the ion with an electron configuration of kr $4d^3$ given it has a charge of +2?
$y^{2+}$
$ti^{2+}$
$tc^{2+}$
$nb^{2+}$
question 16 (0.5 points)
listen
what is the identity of the ion with an electron configuration of ne$3s^2 3p^6$ given it has a charge of -2?
$o^{2-}$
$s^{2-}$
$ar^{2-}$
$mg^{2-}$
Question 15
Step1: Find neutral atom electron count
A +2 ion has 2 fewer electrons than its neutral atom. The ion has electron count: $36$ (from [Kr]) $+3=39$. Neutral atom electrons: $39+2=41$.
Step2: Match to element
Element with atomic number 41 is Niobium (Nb). So the ion is $\text{Nb}^{2+}$.
Question 16
Step1: Find neutral atom electron count
A -2 ion has 2 more electrons than its neutral atom. The ion has electron count: $10$ (from [Ne]) $+2+6=18$. Neutral atom electrons: $18-2=16$.
Step2: Match to element
Element with atomic number 16 is Sulfur (S). So the ion is $\text{S}^{2-}$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Question 15: D. $\text{Nb}^{2+}$
Question 16: B. $\text{S}^{2-}$