QUESTION IMAGE
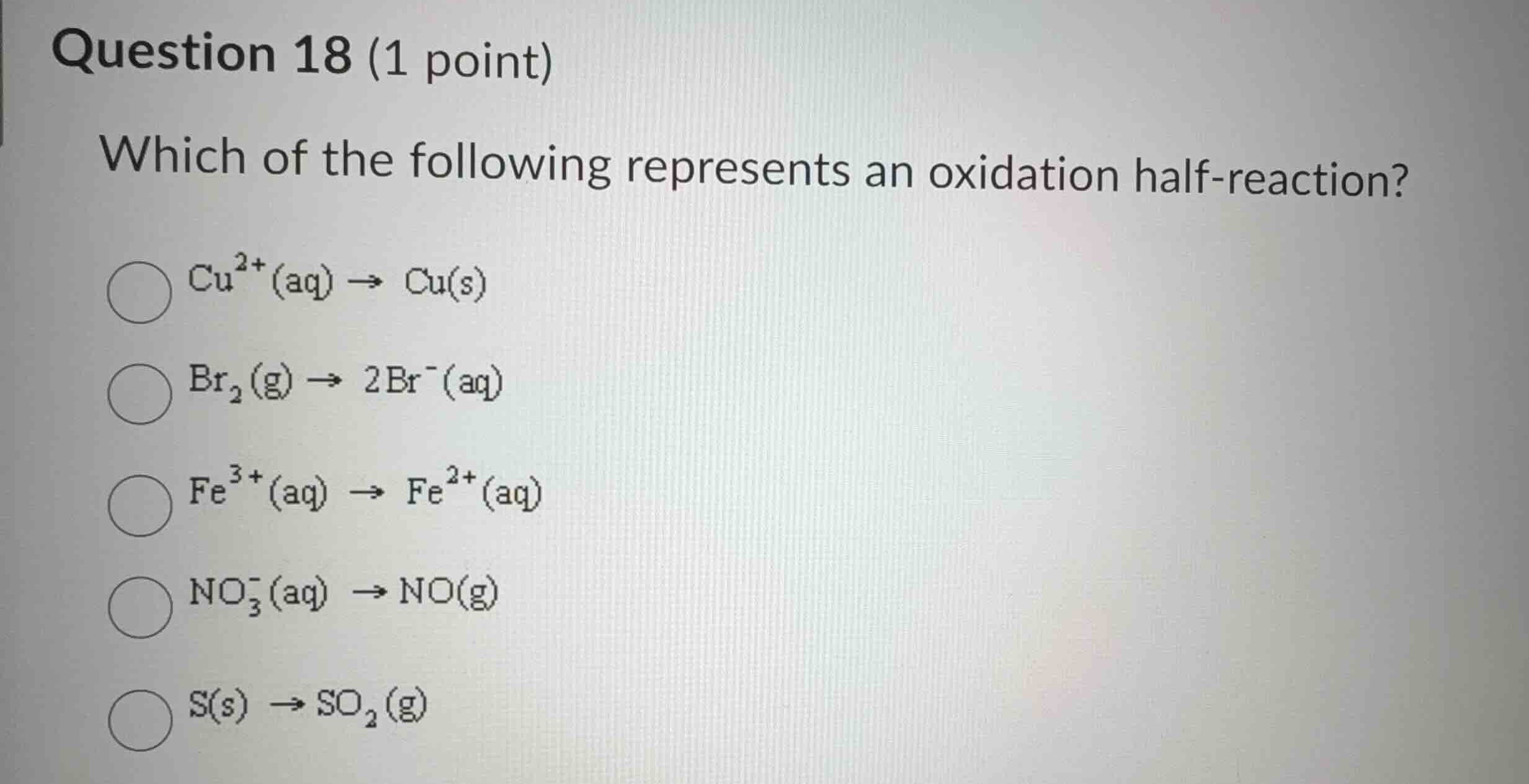
Question
question 18 (1 point)
which of the following represents an oxidation half-reaction?
$\ce{cu^{2+}(aq) \
ightarrow cu(s)}$
$\ce{br_{2}(g) \
ightarrow 2br^{-}(aq)}$
$\ce{fe^{3+}(aq) \
ightarrow fe^{2+}(aq)}$
$\ce{no^{-}_{3}(aq) \
ightarrow no(g)}$
$\ce{s(s) \
ightarrow so_{2}(g)}$
To determine an oxidation half - reaction, we use the concept of oxidation states (oxidation numbers). Oxidation is defined as an increase in the oxidation state (loss of electrons).
- For the reaction $\ce{Cu^{2+}(aq) -> Cu(s)}$: The oxidation state of $\ce{Cu}$ in $\ce{Cu^{2+}}$ is + 2, and in $\ce{Cu(s)}$ it is 0. The oxidation state decreases, so this is a reduction reaction.
- For the reaction $\ce{Br_{2}(g) -> 2Br^{-}(aq)}$: The oxidation state of $\ce{Br}$ in $\ce{Br_{2}}$ is 0, and in $\ce{Br^{-}}$ it is - 1. The oxidation state decreases, so this is a reduction reaction.
- For the reaction $\ce{Fe^{3+}(aq) -> Fe^{2+}(aq)}$: The oxidation state of $\ce{Fe}$ in $\ce{Fe^{3+}}$ is + 3, and in $\ce{Fe^{2+}}$ it is + 2. The oxidation state decreases, so this is a reduction reaction.
- For the reaction $\ce{NO_{3}^{-}(aq) -> NO(g)}$: Let the oxidation state of $\ce{N}$ in $\ce{NO_{3}^{-}}$ be $x$. We know that the oxidation state of $\ce{O}$ is - 2. So, $x + 3\times(-2)=-1$, which gives $x = + 5$. In $\ce{NO}$, let the oxidation state of $\ce{N}$ be $y$. Then $y+(-2)=0$, so $y = + 2$. The oxidation state of $\ce{N}$ decreases from + 5 to + 2, so this is a reduction reaction.
- For the reaction $\ce{S(s) -> SO_{2}(g)}$: The oxidation state of $\ce{S}$ in $\ce{S(s)}$ is 0. In $\ce{SO_{2}}$, let the oxidation state of $\ce{S}$ be $z$. Then $z + 2\times(-2)=0$, so $z= + 4$. The oxidation state of $\ce{S}$ increases from 0 to + 4, which means $\ce{S}$ is oxidized. So this is an oxidation half - reaction.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$\boldsymbol{\ce{S(s) -> SO_{2}(g)}}$ (the last option: S(s) → SO₂(g))