QUESTION IMAGE
Question
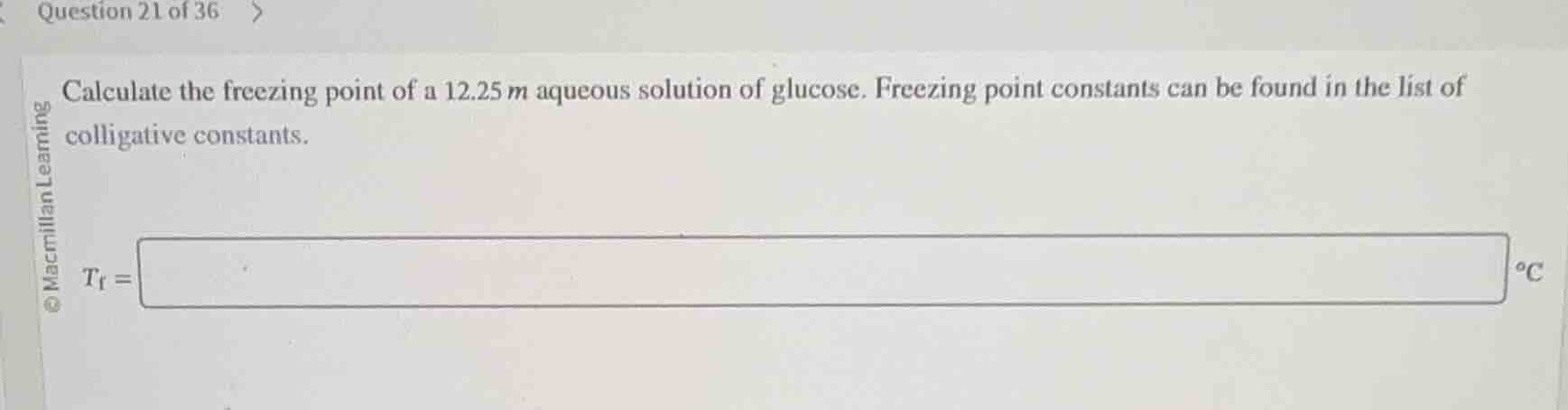
question 21 of 36
calculate the freezing point of a 12.25$m$ aqueous solution of glucose. freezing point constants can be found in the list of colligative constants.
$t_f =$ $\boldsymbol{square}$ $^\circ c$
Step1: Recall freezing point depression formula
The freezing point depression is given by $\Delta T_f = i \times K_f \times m$, where:
- $i$ = van't Hoff factor (for glucose, a non-electrolyte, $i=1$)
- $K_f$ = freezing point depression constant for water, $K_f = 1.86\ ^\circ\text{C}/m$
- $m$ = molality of the solution
Step2: Calculate $\Delta T_f$
Substitute the known values:
$\Delta T_f = 1 \times 1.86\ ^\circ\text{C}/m \times 12.25\ m$
$\Delta T_f = 22.785\ ^\circ\text{C}$
Step3: Find solution freezing point
Pure water freezes at $0^\circ\text{C}$. The solution freezing point $T_f$ is:
$T_f = T_{f,pure} - \Delta T_f$
$T_f = 0^\circ\text{C} - 22.785^\circ\text{C}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$-22.79^\circ\text{C}$ (rounded to two decimal places, or $-22.8^\circ\text{C}$ for reasonable precision)